SYNTHESIS AND CHARACTERIZATION OF GADOLINIUM-BASED NANOCRYSTALS FOR THEIR POTENTIAL APPLICATION AS TRIMODAL IMAGING CONTRAST AGENTS

- lanthanide,
- nanocrystals,
- trimodal,
- gadolinium,
- contrast agents
Copyright (c) 2020 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
Abstract
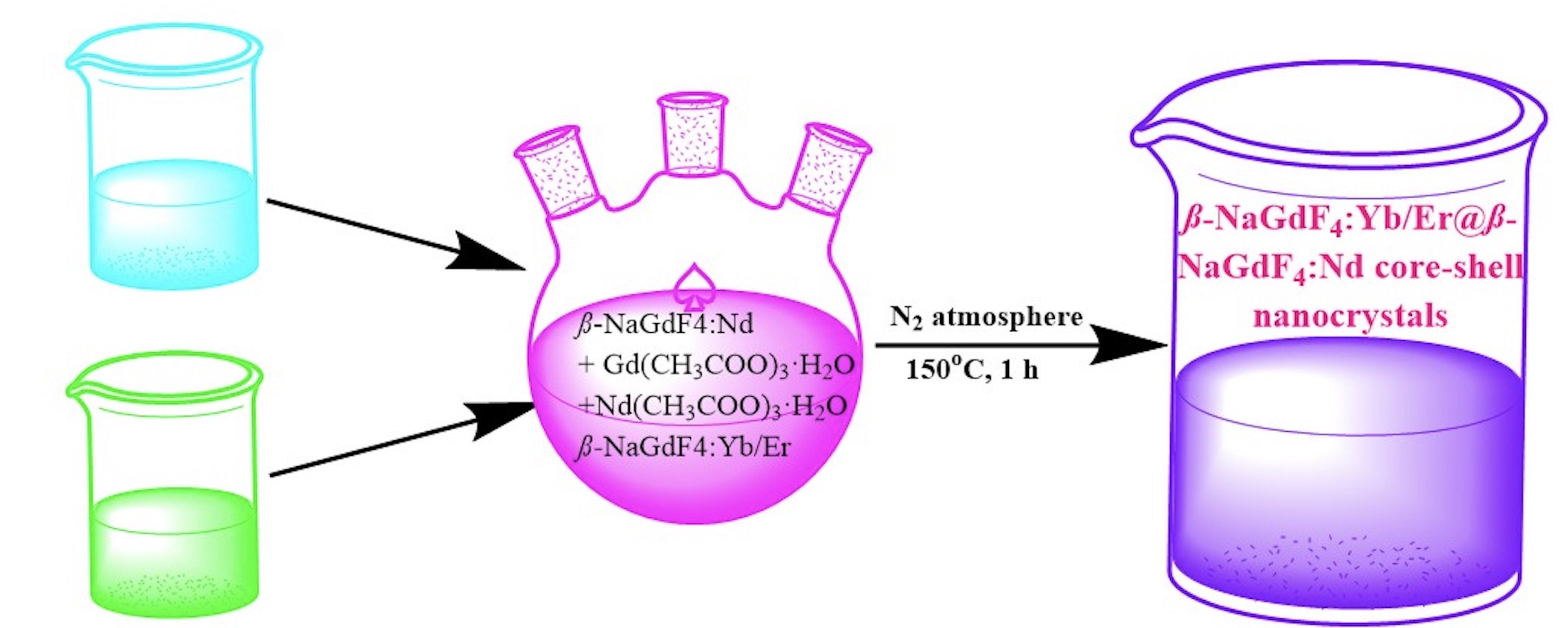
Lanthanide-doped gadolinium nanomaterials have attracted considerable attention due to their promising applications in biological imaging. Sodium gadolinium fluoride (β-NaGdF4) nanomaterials act as perfect host for doping of luminescent lanthanide ions. Due to heavy nature of gadolinium, it can strongly absorb X-ray radiations. Therefore, Gd-based nanomaterials are applied as contrast agents for X-ray tomography (CT). Presence of seven unpaired electrons in Gd+3 ion, its large magnetic moment and long electronic relaxation time makes it an ideal candidate to enhance water proton relaxation rates. These characteristics make Gd-based nanomaterial useful for their potential application as MRI contrast agents. In this work, sodium gadolinium fluoride is doped with ytterbiumand erbium to produce β-NaGdF4:Yb/Er as core nanocrystals. They are subsequently coated with sodium gadolinium fluoride doped with neodymium (β-NaGdF4:Nd) to produce β-NaGdF4:Yb/Er@β-NaGdF4:Nd core-shell nanocrystals. Powder X‐ray diffraction (XRD) results indicated that β-NaGdF4 (host), β-NaGdF4:Yb/Er (core) and β-NaGdF4:Yb/Er@β-NaGdF4:Nd (core-shell) nanocrystals all exist in hexagonal crystalline phase. While Dynamic Light Scattering (DLS) results indicated that the particle size of the synthesized nanocrystals was in range of 12 to 27 nm. FTIR results confirmed that the synthesized nanocrystals were stabilized by oleic acid. Nanocrystals reported in this work are expected to be useful trimodal contrast agents for photoluminescence, X-ray tomography and Magnetic Resonance Imaging (MRI).
References
- References
- A. K. Buck, S. Nekolla, S. Ziegler, A. Beer, B. J. Krause, K. Herrmann, K. Scheidhauer, H.-J. Wester, E. J. Rummeny M. J. J. N. M. Schwaiger, J. Nucl. Med. 49, 1305 (2008)
- A. Galperin, D. Margel, J. Baniel, G. Dank, H. Biton S. Margel, Biomaterials 28, 4461 (2007)
- O. M. Merkel, D. Librizzi, A. Pfestroff, T. Schurrat, M. Béhé T. Kissel, Bioconj. Chem. 20, 174 (2008)
- B. J. Pichler, H. F. Wehrl M. S. J. J. o. N. M. Judenhofer, J. Nucl. Mater. 49, 5S (2008)
- B. J. Pichler, A. Kolb, T. Nägele H.-P. Schlemmer, J. Nucl. Med. 51, 333 (2010)
- R. Bar-Shalom, N. Yefremov, L. Guralnik, D. Gaitini, A. Frenkel, A. Kuten, H. Altman, Z. Keidar O. Israel, J. Nucl. Med. 44, 1200 (2003)
- A. Dirksen, S. Langereis, B. F. de Waal, M. H. van Genderen, T. M. Hackeng E. Meijer, Chem. Commun. 2811 (2005)
- J.-L. Bridot, A.-C. Faure, S. Laurent, C. Riviere, C. Billotey, B. Hiba, M. Janier, V. Josserand, J.-L. Coll L. Vander Elst, J. Am. Chem. Soc. 129, 5076 (2007)
- J. Liu, W. Bu, S. Zhang, F. Chen, H. Xing, L. Pan, L. Zhou, W. Peng J. Shi, Chem. A Eur. J. 18, 2335 (2012)
- S. Adams, R. P. Baum, T. Stuckensen, K. Bitter G. Hör, Eur. J. Nucl. Med. 25, 1255 (1998)
- H. U. Rashid, K. Yu J. Zhou, J. Struct. Chem. 54, 223 (2013)
- R. H. U., M. N. Umar, K. Khan, M. N. Anjum M. Yaseen, J. Struct. Chem. 55, 910 (2014)
- H. U. Rashid, K. Khan, M. Yaseen, W. Hassan M. N. UMAR, Rom. J. Chem. 59, 27 (2014)
- F. Zhang, in Photon Upconversion Nanomaterials, Springer, 2015, pp. 187.
- J. Cheon J.-H. Lee, Acc. Chem. Res. 41, 1630 (2008)
- W. J. Mulder, A. W. Griffioen, G. J. Strijkers, D. P. Cormode, K. Nicolay Z. A. Fayad, Nanomedicine 2, 307 (2007)
- C. Bremer R. Weissleder, Acad. Radiol. 8, 15 (2001)
- T. Persigehl, W. Heindel C. Bremer, Abdom. Imaging 30, 343 (2005)
- D. Sosnovik R. Weissleder, in Imaging in Drug Discovery and Early Clinical Trials, Springer, 2005, pp. 83.
- F. Liu, X. He, L. Liu, H. You, H. Zhang Z. Wang, Biomaterials 34, 5218 (2013)
- D. K. Chatterjee, M. K. Gnanasammandhan Y. Zhang, Small 6, 2781 (2010)
- J. Zhou, X. Zhu, M. Chen, Y. Sun F. Li, Biomaterials 33, 6201 (2012)
- C. Wang, H. Tao, L. Cheng Z. Liu, Biomaterials 32, 6145 (2011)
- K. W. Krämer, D. Biner, G. Frei, H. U. Güdel, M. P. Hehlen S. R. Lüthi, Chem. Mater. 16, 1244 (2004)
- M. He, P. Huang, C. Zhang, H. Hu, C. Bao, G. Gao, R. He D. Cui, Adv. Funct. Mater. 21, 4470 (2011)
- S. Zeng, M.-K. Tsang, C.-F. Chan, K.-L. Wong J. Hao, Biomaterials 33, 9232 (2012)
- Y. F. Wang, L. D. Sun, J. W. Xiao, W. Feng, J. C. Zhou, J. Shen C. H. Yan, Chem. A Eur. J. 18, 5558 (2012)
- S. Dühnen M. Haase, Chem. Mater. 27, 8375 (2015)
- A. Mondry K. J. A. P. P.-S. A. G. P. Bukietynska, Acta Physica Polonica-Series A General Physics 90, 233 (1996)
- J. Tian, X. Zeng, X. Xie, S. Han, O.-W. Liew, Y.-T. Chen, L. Wang X. Liu, J. Am. Chem. Soc. 137, 6550 (2015)
- S. Zheng, W. Chen, D. Tan, J. Zhou, Q. Guo, W. Jiang, C. Xu, X. Liu J. Qiu, Nanoscale 6, 5675 (2014)
- X. Sun Y. Li, Angew. Chem. 116, 607 (2004)
- X. Liu, L. Wang, Z. Wang Z. Li, J. Mater. Res. 26, 82 (2011)

