- TADF,
- pyrazine,
- donor-acceptor,
- RISC,
- TDDFT
Copyright (c) 2019 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
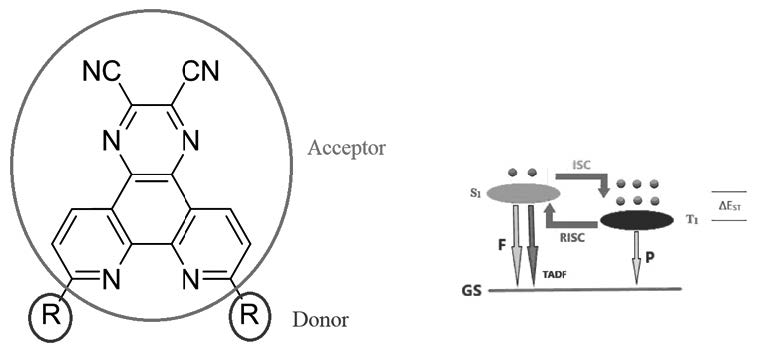
In this study 2,3-dicyanopyrazine based acceptor was combined with donors to obtain Donor-Acceptor type potential thermally activated delayed fluorescence (TADF) emitters (1-10). All molecules’ structural and electronic properties were computed theoretically at the level of Density Functional Theory (DFT) and Time Dependent Density Functional Theory (TDDFT) with the application of three different hybrid functionals. Most of the designed structures have been found to possess the potential to be TADF compounds because they have very narrow energy gap between their first excited singlet and triplet states. As a result, 6-10 molecular pyrazine derivative has been calculated as the best candidate for the purpose. Moreover, having 0.90 eV interfrontier molecular orbital energy band gap, compound 6 has a very strong potential to serve as an efficient OLED material.
References
- N.T. Kalyani, H. Swart, S.J. Dhoble, Principles and Applications of Organic Light Emitting Diodes (OLEDs). Duxford, United Kingdom, Woodhead Printing, 2017.
- G.F. deSa, O.L. Malta, D.C. deMello, A.M. Simas, R.L. Longo, P.A. Santa-Cruz, E.F. daSilva, Spectroscopic properties and design of highly luminescent Lanthanide coordination complexes. Coord. Chem. Rev. 2000, 196, 165–95.
- B.Valeur, M.N. Berberan-Santos, Molecular fluorescence: principles and applications. John Wiley and Sons, 2012.
- S. Basak, K. Chattopadhyay, Studies of protein folding and dynamics using single molecule fluorescence spectroscopy, Phys. Chem. Chem. Phys. 16 (2014) 11139-11149.
- F. Yakuphanoğlu, S. Okur, Analysis of electronic parameters and interface states of boron dispersed triethenolemine/p-Si structure by AFM, I-V, C-V-f and G/w-V-f techniques, Mikroelectronic Engineering, 87 (2010) 30-34.
- Ö.F. Yüksel, N. Tuğluoğlu, H. Şafak, M. Kuş, The motification of Schottky barrier height of Au/p-Si Schottky devices by perylene-diimide, J. Appl. Phys. 113 (2013) 044507-044516.
- S. Okur, F. Yakuphanoğlu, M. Özsöz, P.K. Kadayıfçılar, Electrical and interface properties of Au/DNA/n-Si organic-on-inorganic structures, Microelectr. Eng. 86 (2009) 2305-2311.
- R.K. Gupta, R.A. Singh, Fabrication and characteristics of Schottky diode based on composite organics semiconductor, Comp. Sci. Tech., 65 (2005) 677-681.
- Ş. Aydoğan, Ü. İncekara, A.R. Deniz, A Türüt, Extraction of electronic parameters of Schottky diode based on an organic Orcein, Microelectr. Eng., 87 (2010) 2525-2530.
- Ö. Güllü, S. Asubay, Ş. Aydoğan, A. Türüt, Electrical characterization of the Al/new fuchsin/n-Si organic-modified device, Physica E, 42 (2010) 1411-1416.
- I.S. Yahia, A.A. Farag, F. Yakuphanoğlu, W.A. Farooq, Temperature dependence of electronic parameters of organic Schottky diode based on fluorescein sodium salt, Synthetic Metals, 161 (2011) 881-887.
- P.S. Vincett, W.A. Barlow, R.A. Hann, G.G. Roberts, Electrical conduction and low voltage blue electroluminescence in vacuum-deposited organic films, Thin Solid Films, 94 (1982) 171-183.
- C. Binggeli, Interior Graphic Standards: Student Edition. New Jersey, Wiley, 2012.
- Y.F. Liu, J. Feng, Y.F. Zhang, H.F. Cui, D. Yin, Y.G. Bi, J.F. Song, Q.D. Chen, H.B. Sun, Improved efficiency of indium-tin-oxide-free flexible organic light-emitting devices. Org. Electron. 15 (2014) 478–483.
- Q.Y. Zhang, K. Pita, S. Buddhudu, C.H. Kam, Luminescent properties of rare -earth ion doped yttrium silicate thin film phosphors for a full -colour display, J Phys D 35 (2002) 3085–3090.
- W. Xiaoxiao, L. Fushan, W. Wei, G. Tailiang, Flexible white phosphorescent organic light emitting diodes based on multilayered graphene/PEDOT:PSS transparent conducting film, Appl. Surf. Sci. 295 (2014) 214–218.
- W. Kohn, L.J. Sham, Self-consistent equations including exchange and correlation effects. Phys. Rev. 140 (1965) 1133–1138.
- M.J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox, Gaussian 09, Revision D.01, Gaussian, Inc., Wallingford CT, 2013.
- A.D. Becke, Density-functional exchange-energy approximationwith correct asymptotic behavior, Phys Rev A 1988, 38, 3098–3100.
- C. Lee, W. Yang, R.G. Parr, Development of the Colle–Salvetti correlation energy formula into a functional of the electron density, Phys Rev B 37 (1988) 785–789.
- T. Yanai, D. Tew, and N. Handy, A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP), Chem. Phys. Lett., 393 (2004) 51-57.
- J.-D. Chai and M. Head-Gordon, Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections, Phys. Chem. Chem. Phys., 10 (2008) 6615-6620.
- M.E. Casida, C. Jamorski, K.C. Casida, D.R. Salahub, Molecular excitation energies to high-lying bound states from timedependent density-functional response theory: characterization and correction of the time-dependent local density approximation ionization threshold, J. Chem. Phys. 108 (1998) 4439–4449.
- S.M. Yanez, S.A. Moya, C. Zuniga, G.C. Jiron, Theoretical assessment of TD-DFT applied to a ferrocene-based complex, Comput. Theor. Chem. 1118 (2017) 65–74.
- A. Endo, M. Ogasawara, A. Takahashi, D. Yokoyama, Y. Kato, C. Adachi, Thermally Activated Delayed Fluorescence from Sn4+–Porphyrin Complexes and Their Application to Organic Light Emitting Diodes — A Novel Mechanism for Electroluminescence. Adv. Mater. 21 (2009) 4802–4806.
- A. Endo, K. Sato, K. Yoshimura, T. Kai, A. Kawada, H. Miyazaki, C. Adachi, Efficient up-conversion of triplet excitons into a singlet state and its application for organic light emitting diodes, Appl. Phys. Lett. 98 (2011) 83302-83305.
- R. Czerwieniec, J. Yu, H. Yersin, Blue-Light Emission of Cu(I) Complexes and Singlet Harvesting, Inorg. Chem. 50 (2011) 8293–8301.
- M.J. Leitl, F.R. Küchle, H.A. Mayer, L. Wesemann, H. Yersin, Brightly Blue and Green Emitting Cu(I) Dimers for Singlet Harvesting in OLEDs, J. Phys Chem A 117 (2013) 11823–11836.
- H. Uoyama, K. Goushi, K. Shizu, H. Nomura, C. Adachi, Highly efficient organic light-emitting diodes from delayed fluorescence, Nature 492 (2012) 234–238.
- Z. Khodaee, A. Yahyazadeh, N.O. Mahmoodi, M.A. Zanjanchi, V. Azimi, One-pot synthesis and characterization of new cuprous pyrazinoporphyrazines containing peripherally functionalized units, J. Mol. Struct. 1029 (2012) 92–97.
- J.L. Nishida, S. Murai, E. Fujiwara, H. Tada, M. Tomura, Y. Yamashita, Preparation, Characterization, and FET Properties of Novel Dicyanopyrazinoquinoxaline Derivatives, Org. Lett. 6 (2004) 2007–2010.
- M.A. Ivanov, M.V. Puzyk, K.P. Balashev, Spectroscopic and Electrochemical Properties of Dichlorodiimine Complexes of Au(III) and Pt(II) with 1,4-Diazine Derivatives of o-Phenanthroline, Russ. J. Gen. Chem. 76 (2006) 843–848.
- L. Kozlov and I. Goldberg. Hydrogen-bonding versus [pi]-[pi] stacking interactions in dipyrido[f,h]quinoxaline-6,7-dicarbonitrile and 6,7-dicyanodipyrido[f,h]quinoxalin-1-ium chloride dihydrate, Acta Cryst. 64 (2008) 498-501.
- Z. Turhan Irak, S. Gümüş, Heterotricyclic Compounds via Click Reaction: A Computational Study, Noble Int. J. Sci. Res. 7 (2017) 80-89.
- P. Atkins, T. Overton, J. Rourke, M. Weller, F. Armstrong, Shriver and Atkins Inorganic Chemistry, Fourth Edition, Oxford University Press, Oxford, 2006.
- T. Nakagawa, S.Y. Ku, K.T. Wong, C. Adachi, Electroluminescence based on thermally activated delayed fluorescence generated by a spirobifluorene donor–acceptor structure, Chem. Commun. 48 (2012) 9580–9582.
- G. Mehes, H. Nomura, Q. Zhang, T. Nakagawa, C. Adachi, Enhanced electroluminescence efficiency in a spiro-acridine derivative through thermally activated delayed fluorescence, Angew. Chem. Int. Ed. 51 (2012) 11311–11315.
- K. Nasu, T. Nakagawa, H. Nomura, C.J. Lin, C.H. Cheng, M.R. Tseng, T. Yasuda, C. Adachi, A highly luminescent spiro-anthracenone-based organic light-emitting diode exhibiting thermally activated delayed fluorescence, Chem. Commun. 49 (2013) 10385–10387.
- M.L. Daly, C.A. DeRosa, C. Kerr, W.A. Morris, C.L. Fraser, Blue thermally activated delayed fluorescence from a biphenyl difluoroboron β-diketonate, RSC Adv. 6 (2016) 81631-81635.
- M. Aydemir, G. Haykır, A. Battal, V. Jankus, S.K. Sugunan, F.B. Dias, H. Attar, F. Türksoy, M. Tavaslı, A.P. Monkman, High efficiency OLEDs based on anthracene derivatives: The impact of electron donating and withdrawing group on the performance of OLED, Org. Electr. 30 (2016) 149–157.
- T. Shan, Z. Gao, X. Tang, X. He, Y. Gao, J. Li, X. Sun, Y. Liu, H. Liu, B. Yang, P. Lu, Y. Ma, Highly efficient and stable pure blue nondoped organic light-emitting diodes at high luminance based on phenanthroimidazole-pyrene derivative enabled by triplei-triplet annihilation, Dyes and Pigments 142 (2017) 189–197.
- D. Zhang, C. Zhao, Y. Zhang, X. Song, P. Wei, M. Cai, L. Duan, Highly efficient full-color thermally activated delayed fluorescent organic light-emitting diodes: extremely low efficiency roll-off utilizing a host with small singlet–triplet splitting. ACS App. Mater. Interfaces. 9 (2017) 4769-4777.
- B. Çiçek, Ü. Çalışı, M. Tavaslı, R. Tülek, A. Teke, Synthesis and optical characterization of novel carbazole Schiff bases, J. Mol. Struct. 1153 (2018) 42-47.
- J.A. Seo, M.S. Gong, W. Song, J.Y. Lee, Molecular Orbital Controlling Donor Moiety for High-Efficiency Thermally Activated Delayed Fluorescent Emitters, Chem. Asian J. 11 (2016) 868–873.
- P. Li, Y. Cui, C. Song, H. Zhang, A systematic study of phenoxazine-based organic sensitizers for solar cells, Dyes and Pigments 137 (2017) 12-23.
- A. Gümüş, S. Gümüş, A Computational Study on a Series of Phenanthrene and Phenanthroline Based Potential Organic Photovoltaics, Maced. J. Chem. Chem. Eng. 36 (2017) 239–249.
- N.J. Turro, Modern Molecular Photochemistry, University Science Books, 1991.
- J. Li, Q. Zhang, H. Nomura, H. Miyazaki, C. Adachi, Thermally Activated Delayed Fluorescence from nπ* to nπ* up-Conversion and its Application to Organic Light- Emitting Diodes, Appl. Phys. Lett. 105 (2014) 13301- 13304.
- Y.X. Sun, Q.L. Hao, W.X. Wei, Z.X. Yu, L.D. Lu, X. Wang, Y.S. Wang, Experimental and density functional studies on 4-(3,4-dihydroxybenzylideneamino)antipyrine, and 4-(2,3,4-trihydroxybenzylideneamino)antipyrine, J. Mol. Struct.:Theochem 904 (2009) 74-82.
- C. Andraud, T. Brotin, C. Garcia, F. Pelle, P. Goldner, B. Bigot, A. Collet, Theoretical and experimental investigations of the nonlinear optical properties of vanillin, polyenovanillin, and bisvanillin derivatives, J. Am. Chem. Soc. 116 (1994) 2094-2102.
- V.M. Geskin, C. Lambert, J.L. Bredas, Origin of High Second- and Third- Order Nonlinear Optical Response in Ammonio/Borato Diphenylpolyene Zwitterions: the Remarkable Role of Polarized Aromatic Groups, J. Am. Chem. Soc. 125 (2003) 15651-15658.
- M. Nakano, H. Fujita, M. Takahata, K. Yamaguchi, heoretical Study on Second Hyperpolarizabilities of Phenylacetylene Dendrimer: Toward an Understanding of Structure−Property Relation in NLO Responses of Fractal Antenna Dendrimers, J. Am. Chem. Soc. 124 (2002) 9648-9655.
- D. Sajan, H. Joe, V.S. Jayakumar, J. Zaleski, Structural and electronic contributions to hyperpolarizability in methyl p-hydroxy benzoate, J. Mol. Struct. 785 (2006) 43-53.
- R. Zhang, B. Du, G. Sun, Y.X. Sun, Experimental and theoretical studies on o-, m- and p-chlorobenzylideneaminoantipyrines, Spectrochim. Acta A 75 (2010) 1115-1124.
- D.A. Kleinman, Nonlinear Dielectric Polarization in Optical Media, Phys. Rev. 126 (1962) 1977-1979.
- K.S. Thanthiriwatte, K.M. Nalin de Silva, Non-linear optical properties of novel fluorenyl derivatives—ab initio quantum chemical calculations, J. Mol. Struct.:Theochem 617 (2002) 169-175.
- H. Tanak, K. Pawlus, M.K. Marchewka, A. Pietraszko, Structural, vibrational and theoretical studies of anilinium trichloroacetate: New hydrogen bonded molecular crystal with nonlinear optical properties, Spectrochim. Acta Part A 118 (2014) 82–93.
- E. Scrocco, J. Tomasi, Topics in Current Chemistry, vol. 7, Springer, Berlin, 1973.
- F.J. Luque, J.M. Lopez, M. Orozco, Perspective on Electrostatic interactions of a solute with a continuum. A direct utilization of ab initio molecular potentials for the prevision of solvent effects, Theor. Chem. Acc. 103 (2000) 343–345.
- H. Tanak, A.A. Agar, O. Buyukgungor, Experimental (XRD, FT-IR and UV–Vis) and theoretical modeling studies of Schiff base (E)-N′-((5- nitrothiophen-2-yl)methylene)-2-phenoxyaniline, Spectrochim. Acta Part A 118 (2014) 672–682.
- H. Tanak, Density functional computational studies on 2-[(2,4-Dimethylphenyl)iminomethyl]-3,5-dimethoxyphenol, Int. J. Quant. Chem. 112 (2012) 2392-2402.


