MICROWAVE-ASSISTED LIQUID PHASE MICROEXTRACTION FOLLOWED WITH FLAME ATOMIC ABSORPTION SPECTROMETRY FOR TRACE DETERMINATION OF ZINC IN FOOD SAMPLES
- Microwave-assisted liquid phase microextraction,
- Zinc,
- Atomic absorption spectrometry,
- Food sample
Copyright (c) 2017 Kourosh Motevalli, Zahra Yaghoubi

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
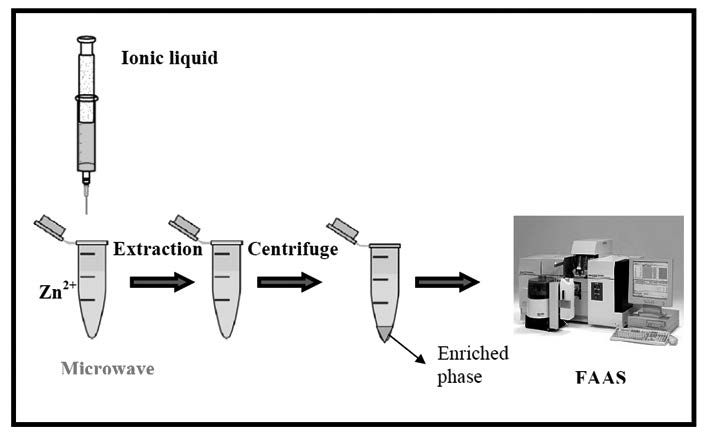
In this work, microwave-assisted liquid phase microextraction (MA-LPME) was followed by flame atomic absorption spectrometry for determination of zinc in food samples. Zinc was complexed with a chelating agent to form hydrophobic species.1-hexyl-3-methylimidazolium hexafluorophosphate ([Hmim][PF6]) ionic liquid (IL) was used as environmentally-friendly extraction solvent. In order to disperse the extraction phase into the sample solution and improve the extraction efficiency, microwave irradiation was used as a disperser agent. The heating effect of microwave irradiation makes the ionic liquid dissolved and dispersed through the sample, which significantly improves the speed and efficiency of extraction. After extraction process, sample solution was centrifuged and the settled phase was diluted and introduced to flame atomic absorption spectrometry by conventional aspiration. Various parameters including amount of ionic liquid, microwave power, irradiation time, pH, amount of chelating agent and salt amount were studied and optimized. At optimized condition, a limit of detection (LOD) of 1.5 ng mL-1, a linear range of 5-32 ng mL-1 and a RSD of 4.3% were obtained. Finally, the developed protocol was utilized for determination of zinc in food samples.
References
- M. J. Khajeh, Hazard. Mater. 172, 385, (2009).
- M. J.Salgueiro, M. B. Zubillaga, A. E. Lysionek,. R. A. Caro, R. Weill, J. R. Boccio, Nutrition 18, 510, (2002).
- J. Borkowska-Burnecka, A. Szymczycha-Madeja, W. Zyrnicki, J. Hazard. Mater. 182, 477, (2010).
- O. Acar, Aanl. Chim. Acta 526,103, (2004).
- J. S. Carletto, K. C. D. P.Roux, H. F. Maltez, E. Martendal, E. Carasek, J. Hazard. Mater. 157, 88, (2008).
- C. Terres-Martos, M. Navarro-Alarcnَ, F. Marin-Lagos, R. Giménez Martinez, De H. L. G. La Serrena, M. C. Lpَez-Martinez, Water Res. 36, 1912, (2002).
- J. A.Salonia, R. G. Wuilloud, J. A. Gsquez, R. A. Olsina, L. D. Martinez, Fresen. J. Anal. Chem. 367, 653, (2000).
- S. Mahesar, S. Sherazi, A. Niaz, M. Bhanger, A. Rauf, Food Chem. Toxicol. 48, 2357, (2011).
- S. S. Saei-Dehkordi, A. A. Fallah, Microchem. J. 98, 156, (2011).
- 10. M. Vega, M. Augusto, M. C. Talio, L. P. Fernindez, Am. J. Anal. Chem. 2, 902, (2011).
- A. Niazi, S. Habibi, M. Ramezani, J. Chil. Chem. Soc. 58, 1899, (2013).
- D. Afzali, M. Fayazi, A. Mostafavi, J. Chil. Chem. Soc. 58, 1593, (2013).
- B. Mokhtari, N. Dalali, K. Pourabdollah, J. Chil. Chem. Soc. 57, 1428, (2012).
- H. Yan, H. Wang, X. Qin, B. Liu, J. Du, J. Pharm. Biomed. Anal. 54, 53, (2011).
- N. Hirayama, M. Deguchi, H. Kawasumi, T. Honjo, Talanta 65, 255, (2005).
- M. Gharehbaghi, F. Shemirani, M. D. Farahani, J. Hazard. Mater. 165, 1049, (2009).
- S. Sun, Y. Wang, W. Z. Yu , T. Q. Zhao, S. Q. Gao, M. Q. Kang, Y. P. Zhang, H. Q. Zhang, Y. Yu, J. Sep. Sci. 34, 1730, (2011).
- L. M. Ravelo-Pérez, J. Hernández-Borges, A. V.; Herrera-Herrera, M. A. Rodríguez- Delgado, Anal. Bioanal. Chem. 395, 2387, (2009).
- L. Vidal, A. Chisvert, A. Canals, A. Salvador, Talanta 81, 549, (2010).
- H. Abdolmohammad-Zadeh, G. H. Sadeghi, Talanta 81,778, (2010).
- S. Wang, C. Liu, S. Yang, F. Liu, Food Anal. Methods 6, 481, (2013).
- S. P. Wen, S. H. Zhu, Food Anal. Methods 7, 291, (2014).


