
- Banana peel,
- bio-adsorbent,
- biomass,
- heavy metal removal,
- musa paradisiaca
- water pollution ...More
Copyright (c) 2026 SCHQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
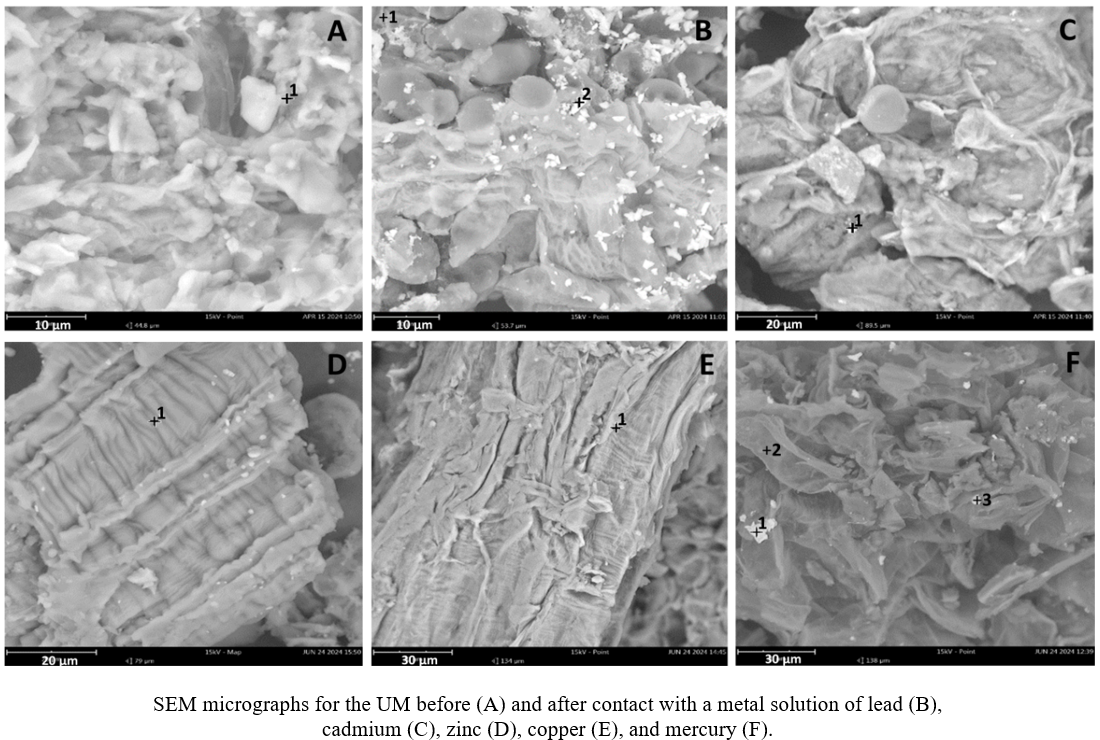
Industrial activities, inadequate waste disposal, and poor control of discharges generated by mining and domestic activities are the primary causes of water pollution with numerous chemical substances, including heavy metals. Several methods have been proposed for removing metals from water, such as precipitation, membrane separation, ion exchange, solid phase extraction, flocculation, and others to address this issue. The metal bio-adsorption using biomass from crops is an alternative that has shown excellent results in recent years. In this work, We studied the bio-adsorption of five metals present in water on biomass from ground banana peel. We evaluated the retention capacity of copper, lead, cadmium, zinc, and mercury on the bio-adsorbent material subjected to acid hydrolysis, basic hydrolysis, and the material without treatment. The material with the best-obtained results was selected to execute a 2k experimental design. The factors were pH, adsorbent dose, and contact time between the adsorbent and the metal ion solution. We used the anodic stripping square wave voltammetry for the mercury determination and adsorptive stripping by square wave voltammetry for copper, zinc, cadmium, and lead quantification. The maximum adsorption capacity ranged from 20 to 30 mg for each gram of material used for the five studied metals. The optimum pH value for mercury was 3.0 while for copper, zinc, cadmium, and lead was 5.0. The optimum contact time was 15 minutes. Mercury is captured differently from the other metals. The materials were characterized using the techniques of Infrared Spectroscopy, Scanning Electron Microscopy, energy-dispersive X-ray Spectroscopy, and thermogravimetric analysis.
References
- Prodipto Bishnu, A., Shafiul Islam, M., Shreejana K. C., Das, A., Anjum, A., Poudel, A., Akter Suchi, A. Heliyon. 10. 1-15. (2024). https://doi.org/10.1016/j.heliyon.2024.e28357
- Rabia Choudhury, T., Acter, T., Abbas Alam, M. Faysal Sowrav, S., Safiur Rahman, M., Sarwaruddin Chowdhury, A. M., Quraishi, S. B. Reg. Stud. Mar. Sci. 70. 1-14. (2024). https://doi.org/10.1016/j.rsma.2024.103378
- Upadhyay, V., Kumari, A., Kumar, S. Chemosphere. 354. 1-9. (2024). https://doi.org/10.1016/j.chemosphere.2024.141697
- Biswal, B. K., Balasubramanian, R. Environ. Chem. Eng. 11 (5). (2023). https://doi.org/10.1016/j.jece.2023.110986
- Malviya, A., Jaspal, D. Resource Recovery in Drinking Water Treatment. 93-105. (2023). https://doi.org/10.1016/B978-0-323-99344-9.00007-4
- Thirunavukkarasu, A., Nithya, R., Sivashankar, R. Chem Eng J. Advances. 8. 1-9. (2021). https://doi.org/10.1016/j.ceja.2021.100188
- Penpun Tasaso. J. Clean Energy Technol. 2, 154-157. (2014). https://doi: 10.7763/JOCET.2014.V2.112
- Tehseen Aman, Asrar Ahmad Kazi, Muhammad Usman Sabri, Qudsia Bano. Colloids Surf. B: Biointerfaces. 63. 116–121 (2008) https://doi:10.1016/j.colsurfb.2007.11.013
- D. Anitha, A. Ramadevi, R. Seetharaman. Mater. Today: Proceedings. 45. 658–662. (2021). https://doi.org/10.1016/j.matpr.2020.02.730
- Ilhem Ghodbane, Oualid Hamdaoui. J. Hazard Mater. 160. 301–309. (2008). https://doi:10.1016/j.jhazmat.2008.02.116
- Litza H. Velazquez-Jimenez, Andrea Pavlick, J. Rene Rangel-Mendez. Ind. Crops Prod. 43. 200– 206. (2013). https://doi.org/10.1016/j.indcrop.2012.06.049
- Pongthipun Phuengphai, Thapanee Singjanusong, Napaporn Kheangkhun, Amnuay Wattanakornsiri. Water Sci. Eng. 14. 286e294. (2021). https://doi.org/10.1016/j.wse.2021.08.003
- Obike A.I., Igwe J.C., Emeruwa C.N., Uwakwe K.J. J. Appl. Sci. Environ. Manage. 22. 182 – 190. (2018). https://doi.org/10.4314/jasem.v22i2.5
- E. Pehlivan, T. Altun, S. Parlayici. Food Chem. 135. 2229-2234. (2012). https://doi.org/10.1016/j.foodchem.2012.07.017
- David Castro, Nelly Ma. Rosas-Laverde, María Belén Aldás, Cristina E. Almeida-Naranjo, Víctor H. Guerrero, Alina Iuliana Pruna. Mater, 14, 2134. (2021). https://doi.org/10.3390/ma14092134
- Hikmatullah Ahmadi, Sayed Sadat Hafiz, Habibullah Sharifi, Ngambua Ngambua Rene, Sayed Sanaullah Habibi, Shakeel Hussain. Case Stud. Chem. Environ. Eng. 6. 100242. (2022). https://doi.org/10.1016/j.cscee.2022.100242
- Nordiana Suhada Mohmad Tahiruddin, Rosmawati Abdul Aziz, Rosliza Ali, Nurul Izza Taib. J. Environ. Chem. Eng. 11. 109953. (2023). https://doi.org/10.1016/j.jece.2023.109953
- Dalal Z. Husein. Desalin Water Treat. 1-9. (2013). https://doi:10.1080/19443994.2013.801793
- E. Khoramzadeh, B. Nasernejad, R. Halladj. J. Taiwan Inst. Chem. Eng. 44. 266–269. (2013). https://doi.org/10.1016/j.jtice.2012.09.004
- Hui Ge, Feng Xie, Shaohua Wu, Wei Wang. J. Clean. Prod. 434. 139926. (2024). https://doi.org/10.1016/j.jclepro.2023.139926
- El Mansouri, N. E. [Thesis]. Universidad de Tarragona. España. (2007).
- Dias, F., Meira, L. A., Carneiro, C. N., Dos Santos, L. F., Guimarães, L. B. Coelho, N. M., Coelho, L. M., Alves, V. N. TrAC Trends Anal. Chem. 158. 1-14. (2023). https://doi.org/10.1016/j.trac.2022.116891
- Sharda Gupta, Dhananjay Kumar, J.P. Gaur. Chem. Eng. J. 148, 226-233. (2009). https://doi.org/10.1016/j.cej.2008.08.019
- KKIU Arunakumara, Buddhi Charana Walpola and Min-Ho Yoon. Korean J. Environ Agric. 32, 108-116. (2020). https://doi.org/10.5338/KJEA.2013.32.2.108
- Kovo G. Akpomie, Jeanet Conradie. Environ. Chem. Lett. 18:1085–1112. (2020). https://doi.org/10.1007/s10311-020-00995-x
- Nan Zhou, Honggang Chen, Junting Xi, Denghui Yao, Zhi Zhou, Yun Tian, Xiangyang Lu. Bioresour. Technol. 232, 204-210. (2017). https://doi: 10.1016/j.biortech.2017.01.074.
- Vinay Kumar, Pritha Chakraborty, Poonam Janghu, Mridul Umesh, Suma Sarojini, Ritu Pasrija, Komalpreet Kaur, Sivarama Krishna Lakkaboyana, Vimal Sugumar, Manivannan Nandhagopal, Azhagu Madhavan Sivalingam. Carbohydr. Polym. Technol. Appl. 6. 100366. (2023). https://doi.org/10.1016/j.carpta.2023.100366
- Aydin, H., Bulut, Y. and Yerlikaya, C. J. Environ. Manag. 87. 37–45. (2008). https://doi: 10.1016/j.jenvman.2007.01.005
- Zümriye A., İşoğlu, I. A. Process Biochem. 40. 3031-3044. 2005; https://doi.org/10.1016/j.procbio.2005.02.004
- E.-S. Z. El-Ashtoukhy, N. K. Amin, O. Abdelwaha. Desalination. 223, 1–3, 162-173. (2008). https://DOI: 10.1016/j.desal.2007.01.206
- Tan, G. & Xiao, D. J. Hazard. Mater. 164. 1359-1363. (2009). https://doi.org/10.1016/j.jhazmat.2008.09.082
- Ding, Y., Jing, D., Gong, H., Zhou, L. & Yang, X. Bioresour. Technol. 114. 20-25. (2012). https://doi: 10.1016/j.biortech.2012.01.110
- Tan, G., Yuan, H., Liu, Y. & Xiao, D. J. Hazard. Mater. 174. No. 1-3. 740-745. (2010). https://DOI: 10.1016/j.jhazmat.2009.09.114
- Wilavan Jaihan, Vanee Mohdee, Sompop Sanongraj, Ura Pancharoen, Kasidit Nootong. Arabian J. Chem. 15, Issue 7, 103883. (2022). https://DOI: 10.1016/j.arabjc.2022.103883
- Zhiyuan Liu, Feng Zhen, Quanguo Zhang, Xin Qian, Wenzhe Li, Yong Sun, Lingling Zhang, Bin Qu. Bioresour. Technol. 359, 127471. (2022). https://doi.org/10.1016/j.biortech.2022.127471.
- Khoramzadeh, E., Nasernejad, B., Halladj, R. J Taiwan Inst. Chem. Eng. 44, 266–269. (2013). https://doi.org/10.1016/j.jtice.2012.09.004
- Fiol, N., Villaescusa, I., Martínez, M., Miralles, N., Poch, J., Sep. Purif. Technol. 50. 132–140. (2005). https://doi:10.1016/j.seppur.2005.11.016
- Lekan Taofeek Popoola , Adeyinka Sikiru Yusuff, Abel Adekanmi Adeyi, Oluwagbenga Olawale Omotara. South African J. Chem. Eng. 39 19–27. (2022). https://DOI: 10.1016/j.sajce.2021.11.001.
- Boonamnuayvitaya, V., Chaiya, C., Tanthapanichakoon, W., Jarudilokkul, S. Separ. Purif. Technol. 35. 11–22. (2003). https://doi:10.1016/S1383-5866(03)00110-2
- Sousa, F. W., Oliveira, A. G., Ribeiro, J. P., Rosa, M. F., Keukeleire, D. & Nascimento, R. F. J. Environ. Manag. 91. No. 8. 1634-1640. (2010). https://DOI: 10.1016/j.jenvman.2010.02.011
- Meringer, A., Liffourrena, A. S., Heredia, R. M., Lucchesi, G. I. and Boeris, P. S. J. Biotechnol. vol. 328, 87–94. (2021). https://DOI: 10.1016/j.jbiotec.2021.01.011
- G.C. Panda, S.K. Das, A.K. Guha. Colloids Surf. B: Biointerfaces. 62, 173-179. (2008). https://doi: 10.1016/j.colsurfb.2007.09.034
- Velazquez-Jimenez, L. H., Pavlick, A., Rangel-Mendez, J. R. Ind. Crops Prod. 43, No. 1, 200–206. (2013). https://DOI: 10.1016/j.indcrop.2012.06.049
- Xiaomin Li, Yanru Tang, Xiuju Cao, Dandan Lu, Fang Luo, Wenjing Shao. Colloids Surf. A: Physicochem. Eng. Asp. 317, 512-521. (2008). https://doi: 10.1016/j.colsurfa.2007.11.031
- Geoffrey S. Simate, Sehliselo Ndlovu. J. Ind. Eng. Chem. 21. 635-643. (2015). https://DOI: 10.1016/j.jiec.2014.03.031
- Rocha, C. G., Zaia, D. A., Alfaya, S. J. Hazard. Mater. 166, 383–388. (2009). https://DOI: 10.1016/j.jhazmat.2008.11.074
- Tian Z., Chen X., Wang Q., and Yang G. Bioresources. 12, 2609–2617. (2017). https://DOI: 10.15376/biores.12.2.2609-2617
- Wade, L. C., Simek J. Organic Chemistry. 9th edition. Pearson. Prentice-Hall. (2020).

