ECOTOXICOLOGICAL EVALUATION OF Lippia alba Mill PLANT RESIDUES ON TERRESTRIAL ECOSYSTEM REPRESENTATIVES
- Lippia alba,
- plant residues,
- ecotoxicology,
- soil respiration,
- soil amendment
- Phaseolus vulgaris,
- phytotoxicity,
- vermicomposting ...More
Copyright (c) 2026 SCHQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
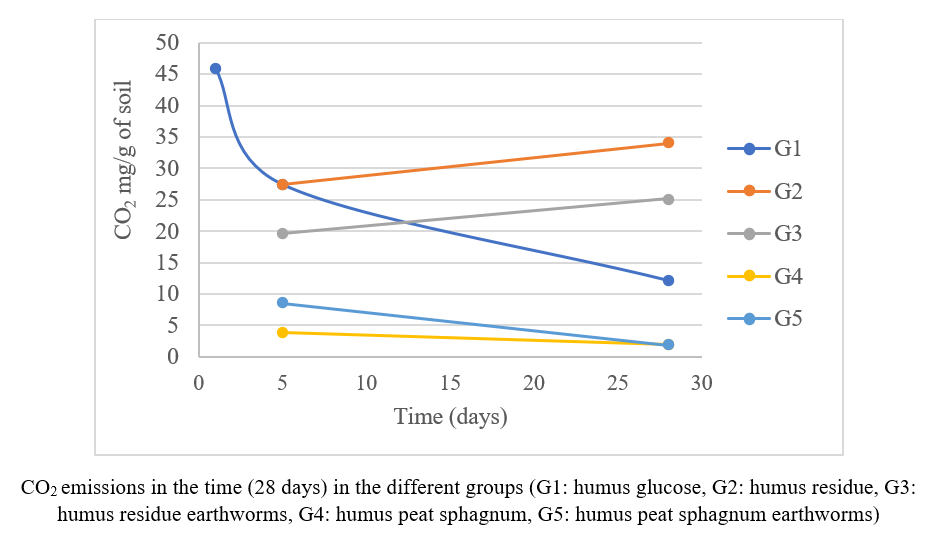
Lippia alba is a medicinal plant commonly used in Cuba and the Caribbean for thedevelopment of herbal formulations against skin inflammatory illnesses thus generating residual biomass whose environmental impact remains unassessed. This study aimed to evaluate the ecotoxicological effects of L. alba residues and their aqueous extract fractions on key terrestrial ecosystem organisms. Soil microbial community activity was analyzed through CO₂ emissions and ammonium (NH₄⁺) quantification, as well as degradation and toxicity tests on earthworms (Eisenia andrei). Phytotoxicity was assessed using Phaseolus vulgaris seeds to determine germination rates and radicle length. Results showed that CO₂ emissions increased in the groups treated with L. alba residues, suggesting enhanced microbial respiration, whereas NH₄⁺ levels remained unchanged across all treatments. Seed germination rates remained above 75% at all tested extract concentrations except at 75%. Radicle length, however, was significantly reduced at higher concentrations (75% and 100%).No toxic effects were observed in E. andrei (earthworms). These findings indicate that L. alba residues are not toxic to soil macro- or microfauna and may contribute to soil organic enrichment, although higher concentrations of aqueous extracts exhibit moderate phytotoxic effects. The study supports the potential incorporation of L. alba waste into sustainable soil management strategies, such as composting and vermicomposting.
References
- S. Alfaro-Araya and R. Díaz, Antioxidant activity and phytoactive compounds related to biological effects present in native southern chilean plants: a review, J. Chil. Chem. Soc., vol. 69, no. 2, pp 6115-6128, (2024).
- S. Nara, R.C. Pascon, M.A. Vallim, C.R. Figueiredo, M.G. Soares, J.H. Lago, et al., Cytotoxic and antimicrobial constituents from the essential oil of Lippia alba (Verbenaceae), Medicines, vol. 3, no. 3, pp. 22, (2016), doi: 10.3390/medicines3030022.
- S. Malik, S. Odeyemi, G.C. Pereira, Jr. L. Mamede de Freitas, H. Abdul-Hamid, N. Atabaki, et al., New insights into the biotechnology and therapeutic potential of Lippia alba (Mill.) N.E.Br. ex P. Wilson, J Essent. Oil Res., vol. 33, no. 6, pp 523-535, (2021), doi:10.1080/10412905.2021.1936667.
- C. Chaverri, F. Ramón-Farías, J.F. Cicció, Chemistry of essential oils of the shrub Lippia alba (Verbenaceae) from Mexico and Costa Rica, UNED Res. J., vol. 14, no. 2, (2022), doi: 10.22458/urj.v14i2.4005
- D. Cruz, Formulario Nacional de Fitofármacos y Apifármacos, Segunda edición. ECIMED: La Habana, MINSAP, pp131-133, (2017).
- L.B. Pereira, Y.I. Gutiérrez, A. Sánchez, V. García, G. García, Lippia alba (Mill.), una alternativa para nuestra medicina natural y tradicional. Tesis en opción al título de Licenciado en Ciencias Farmacéuticas. Instituto de Farmacia y Alimentos, Universidad de La Habana, (2017).
- C.M. Pérez, A.G. Michaluk, C.A. Torres, V. Mouriño, D.A. Chiappetta, M.B. Núez, Influence of herbal extracts in physicochemical properties and stability of antibacterial gel, J. Adv. Pharm. Educ. Res., vol. 13, no. 4, pp. 16-24, (2023), doi:10.51847/hUu6HHPZZa.
- A.F. Gomes, M.P. Almeida, A.L.M. Ruela, J.G. Amaral, J.M. David et al., Development and evaluation of physical and release properties of a tablet formulation containing dry hydroethanolic extract from Lippia alba leaves. J. Herbal Med.; vol. 29,(October 2021). doi: 10.1016/j.hermed.2021.100459.
- M. Prates de Almeida, L. Alves de Oliveira, L. Almeida et al., Qualitative and quantitative characterization of spray-dried extracts and the development of an innovative oral solid formulation from Lippia alba. Separation Science Plus.; vol. 6, no. 4,(2023). doi:10.1002/sccp.202200076.
- D. Das, N. Kalita, D. Langthasa, V. Faihriem, G. Borah, P. Chakravarty, H. Deka. Eisenia fetida for vermiconversion of waste biomass of medicinal herbs: Status of nutrients and stability parameters, Bioresour Technol, 347:126391. (2022 Mar), doi: 10.1016/j.biortech.2021.126391.
- M.K. Abbasi, M. Hina, A. Khalique y S.R. Khan, Mineralization of three organic manures used as nitrogen source in a soil incubated under laboratory conditions. Comm. Soil Sci. Plant Anal., vol. 38, pp. 1691-1711, (2007), doi:10.1080/00103620701435464.
- OECD (1984). Earthworm Acute Toxicity Tests. Test No. 207.
- EPA (2012). Ecological Effects Test Guidelines: Soil Microbial Community Toxicity Test. OCSPP 850.3200.
- M.R. Moitinho, M.P. Padovan, A.R. Panosso, D. de B. Teixeira, A. S. N. Ferraudo, La Scala J. On the spatial and temporal dependence of CO2 emission soil properties in sugarcane (Saccharum sp.) production, Soil and Tillage Res., pp. 127–132, (2015).
- Z. He & H. Zhang, Applied manure and nutrient chemistry, Springer, (2014), doi:10.1007/978-94-017-8807-6.
- Z. González-Giro, P.L. Batista-Corbal, Y. González-Pérez, E. Rodríguez-Leblanch, E. Marcos-Albear, Evaluación de la fitotoxicidad de un extracto acuoso del alga Padina
- gymnospora (Kützing) sobre semillas de Lactuca sativa L. Biotecnología Vegetal. Instituto de Biotecnología de las Plantas. UCLV. MES, vol. 18, no. 3, pp. 181 – 188, (julio - septiembre 2018).
- P. Filannino, Y. P. Bai, R. Di Cagno, M. Gobbetti, M. G. Gänzle, Metabolism of phenolic compounds by Lactobacillus sp during fermetation of cherry juice and broccoli puree, Food Microbiol., pp. 272-279, (2015), Available at:
- 1016/j.fm.2014.08.018.
- A. Balkrishna, S. Srivastava, D. Srivastava, N. Sharma, V. Arya, A. K.Gautam, Unleashing the potential of medicinal and aromatic plant wastes with particular consideration of vermicomposting: A comprehensive review of literatura. J. Appl Res. Med. Aromat.
- C. García-Gutiérrez, J.A. Félix-Herrán, A. Cárdenas-Flores, B.E. Gómez-Luna, G.M. Ruiz-Aguilar, Técnicas de caracterización de suelos y abonos orgánicos. Fundación Produce Sinaloa, (2014).
- M.M.S. Moreira y J.O. Siqueira, Microbiologia e bioquímica do solo. Ed. UFLA. Brasil. (2002).
- J.P. Anderson, Soil respiration. In: Page AL, Miller RH, Keeney DR (eds). Methods of soil analysis: Chemical and Microbiological properties. 2nd ed. American Society of Agronomy Inc, Wisconsin, USA, pp. 831 -87, (1982).
- A.L. Page, R.H. Miller y D.R. Keeney, Methods of soil analysis. Part 2. Chemical and microbiological properties. Second edition. A. L. Page, R. H. Miller and D. R. Keeney, editors. American Society of Agronomy, Inc. Madison, Wisconsin, USA. (1982).
- OECD 208. Early Seedling Growth Toxicity Test. Ecological Effects Test Guidelines. OECD guidelines for testing of chemical Paris, France. (1996).
- Environmental Protection Agency (EPA): Early Seedling Growth Toxicity Test. Ecological Effects Test Guidelines. OPPTS 850.4230. EPA 712–C–010. (1996).
- T. Poblete, K. Rebolledo, C. Barrera, D. Ulloa, M. Valenzuela, C. Valenzuela, E Pavez, R. Mendoza, C. Narbona, J. González, S. Estevez, R. Ortega and C.González, Effect of germination and cooking on iron content, phytic acid and lectins of four varieties of chilean beans (Phaseolus vulgaris), J. Chil. Chem. Soc., vol. 65, no. 4, pp. 4937-4942, (2020).
- International Business Machines Corporation. SPSS Statistics for Windows [software]. Version 22, (2013).
- R. Fuentes, Agrosistemas sostenibles y ecológicos: la reconversión agropecuaria. Universidad Santiago de Compostela, pp. 250, (2007).
- E. Suárez, S. Chimbolema & R. Jaramillo, Turberas de páramo en el Ecuador: Notas sobre la ecología, conservación, y restauración de un ecosistema estratégico. Imprenta Don Bosco, Quito. 2022;124 p.
- J.C. Benavides, Perturbaciones en las turberas de páramo: la acción del hombre y el clima. En: Visión socioecosistémica de los páramos y la alta montaña colombiana. Instituto de Investigación de Recursos Biológicos Alexander Von Humboldt. Bogotá, D.C. Colombia, (2013).
- J.A. Laynez, J.R. Méndez, Efectos alelopáticos de extractos acuosos de hojas de botón de oro [Tithonia diversifolia (Hemsl.) A. Gray.] sobre la germinación de semillas y crecimiento de plántulas de lechuga (Lactuca sativa L.), Scientia Agropecuaria.; vol. 4, pp. 229-241, (2013), disponible en: www.sciagropecu.unitru.edu.pe.
- L.É. Coelho, S.M. de Oliveira, L.A. de Souza, L. Hernandez- Pastorini, Phytotoxic effects of Aeschynomene fluminensis Vell. on the initial growth of weeds and cultivated plants, Res. Soc. Developmen, vol.10 no. 2, (2021), doi: 10.33448/rsd-v10i2.12551.
- L. Beesley, Respiration (CO2 flux) from urban and peri-urban soils amended with green waste compost. Geoderma., pp. 68–72,(2014), doi: 10.1016/j.geoderma.2014.01.024.
- D. Serri, V. Faggioli, C. Lorenzon, Soil Phosphorus: Stubble quality and microbial descomposition under contrasting water contents fosforo suelo. Ciencia del Suelo.; vol. 35 no. 2, pp. 239-248, (2017).
- A.C. Noa-Rodríguez, Y. Domínguez, Y.I. Gutiérrez, O.C. Beiro, M. Díaz, R. Scull, J.C. Castillo, A. Felipe. Evaluación ecotoxicológica de los residuos de Lippia alba Mill. sobre Eisenia andrei (lombriz de tierra), Rev. Med. Militar., vol. 52, no. 3:e02302950, (2023), Disponible en: https://revmedmilitar.sld.cu.
- E. Monsalve, R. Muñoz-Arriagada, N. Bahamonde, & O.J. Vidal, Caracterización ecológica de una turbera ombrogénica en Magallanes: hacia una propuesta de bioindicadores de monitoreo ambiental. Gayana. Botánica, vol. 78, no. 1, pp. 38-55. (2021), doi:10.4067/S0717-66432021000100038.
- F.A. Einhellig y col., The physiology of allelochemical action: Clues and views. FirstEuropeanAllelopathySymposium. Vigo, España., pp. 3-25, (2002).
- L. Nandakumar and N. S. Rangaswamy. Effect of some Flavonoids and Phenolic Acids on Seed Germination and Rooting. Department of Botany, University of Delhi, India, J. Experimental Botany., vol. 36, no. 169: pp. 1313-1319, (August 1985).
- N. Chaves, J.L. Ríos, C. Gutiérrez, J.C. Escudero, y J.M. Olías, Analysis of secreted flavonoids of Cistus ladanifer L. by high-performance liquid chromatography-particle beam mass spectrometry, J. Chrom. A., pp. 111-115, (1998).
- S. Turab-Raza, B. Zhu, Z. Yao, J. Wu, Z. Chen, Z. Ali, J. Tang, Impacts of vermicompost application on crop yield, ammonia volatilization and greenhouse gases emission on upland in Southwest China, Sci. Total Environment, (2023) doi:10.1016/j.scitotenv.2022.160479.


