- Brassinosteroids, analogs, 23,24-dinorcholane, synthesis.
Copyright (c) 2026 SCHQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
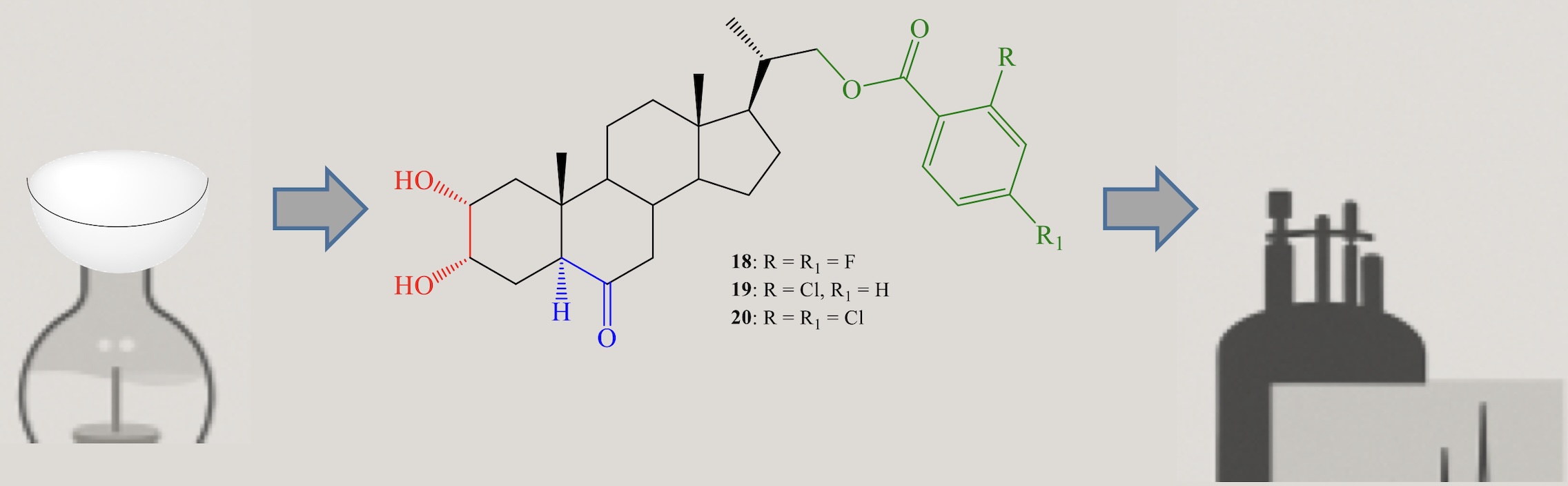
Brassinosteroids are an important family of plant hormones involved in various processes of plant growth and development. They also play a crucial role in plant stress responses, improving tolerance to abiotic factors such as temperature extremes, drought, and salinity, and contributing to resistance against biotic stresses. This work describes the synthesis and full structural characterization of three new 23,24-dinorcholane-type brassinosteroid analogs bearing benzoate groups at C-22 and substituted with fluorine and chlorine atoms, while maintaining the structural features of the A/B rings present in active natural brassinosteroids such as castasterone. The synthesis was achieved in two steps: an esterification reaction with acyl chlorides, followed by a stereospecific Sharpless dihydroxylation. Both reactions proceeded with good yields.
References
- Bajguz, A. Brassinosteroids-Occurrence and chemical structures in plants. In Brassinosteroids: A Class of Plant Hormone. Hayat, S., Ahmad, A., Eds.; Springer: Dordrecht, The Netherlands, pp. 1-27, 2016.
- Nolan, T. M.; Vukašinović, N.; Liu, D.; Russinova, E.; Yin, Y. Brassinosteroids: Multidimensional Regulators of Plant Growth, Development, and Stress Responses. Plant Cell 2020, 32(2), 295-318.
- Li, S.; Zheng, H.; Lin, L.; Wang, F.; Sui, N. Roles of brassinosteroids in plant growth and abiotic stress response. Plant Growth Regul. 2021, 93(1), 29-38.
- Hafeez, M. B.; Zahra, N.; Zahra, K.; Raza, A.; Batool, A.; Shaukat, K.; Khan, S. Brassinosteroids: Molecular and physiological responses in plant growth and abiotic stresses. Plant Stress 2, 2021, 100029.
- Aryal, D. and Alferez, F. Brassinosteroids: Biosynthesis, Signaling, and Hormonal Crosstalk as Related to Fruit Yield and Quality. Plants 2025, 14(12), 1865.
- Thompson M. J.; Meudt W. J.; Mandava N. B.; Dutky S. R.; Lusby W. R.; Spaulding D. W. Synthesis of Brassinosteroids and Relationship of Structure to Plant Growth-Promoting Effects. Steroids 1982, 39(1) 89-105.
- Takatsuto S.; Yazawa N.; Ikekawa N.; Morishita T.; Abe H. Synthesis of (24R)-28-homobrassinolide analogues and structure-activity relationships of brassinosteroids in the rice-lamina inclination test. Phytochemistry 1983, 22(6), 1393-1397.
- Takatsuto, S.; Yazawa, N.; Ikekawa, N.; Takematsu, T.; Takeuchi, Y.; Koguchi, M. Structure Activity Relationship of Brassinosteroids. Phytochemistry 1983, 22(11), 2437-2441.
- Takatsuto, S.; Ikekawa, N.; Morishita, T.; Abe, H. Structure Activity Relationship of Brassinosteroids with Respect to the A/B-Ring Functional-Groups. Chem. Pharm. Bull. 1987, 35(1), 211-216.
- Brosa, C.; Capdevila, J. M.; Zamora, I. Brassinosteroids: A new way to define the structural requirements. Tetrahedron 1996, 52(7), 2435-2448.
- Zullo M. A. T.; Adam G. Brassinosteroid phytohormones: Structure, bioactivity and applications. Braz. J. Plant Physiol. 2002, 14(3), 143-181.
- Hayat, S. & Ahmad, A. Brassinosteroids Bioactivity and Crop Productivity. 1st edition. Dordrecht: Springer, 2003.
- Kovganko, N. V.; Ananich, S. K. Advances in the chemical synthesis of brassinosteroids. Chem. Nat. Compd. 1997, 33(4), 389-416.
- Khripach, V. A.; Zhabinskii, V. N.; de Groot, A. E. Basic synthetic methods and formal syntheses. In Brassinosteroids. A New Class of Plant Hormones. Eds. Academic Press: San Diego, Chapter VI, pp 73-135, 1999.
- Khripach, V.; Zhabinskii, V.; Ermolovich, Y. Synthetic Aspects of Brassinosteroids. Studies in Natural Products Chemistry, Atta-ur-Rahman FRS, University of Karachi, Karachi, Pakistan, Chapter 6, 44, pp. 309-352, 2015.
- Nuñez, M.; Wang, Y.; Russinova, E.; Estévez-Braun, A.; Amesty, A.; Olea, A. F.; Mellado, M.; Díaz, K.; Espinoza-Catalán, L. Synthesis, Biological Activity, and Molecular-Docking Studies of New Brassinosteroid Analogs. Int. J. Mol. Sci. 2024, 25(18), 10158.
- Aitken, V.; Diaz, K.; Soto, M.; Olea, A. F.; Cuellar, M. A.; Nuñez, M.; Espinoza-Catalán, L. New Brassinosteroid Analogs with 23,24-Dinorcholan Side Chain, and Benzoate Function at C-22: Synthesis, Assessment of Bioactivity on Plant Growth, and Molecular Docking Study. Int. J. Mol. Sci. 2024, 25(1), 419.
- Núñez, M. Síntesis de nuevos análogos de brasinoesteroides con cadena lateral del tipo 23,24-bisnorcolánicos, benzoilados en C-22 y su evaluación como reguladores de crecimiento de plantas. Tesis de Doctorado, Departamento de Química, Universidad Técnica Federico Santa María, Chile. 2024.
- Jorquera, S.; Soto, M.; Díaz, K.; Nuñez, M.; Cuellar, M. A.; Olea, A. F.; Espinoza-Catalán L. Novel Brassinosteroid Analogues with 3,6 Dioxo Function, 24-Nor-22(S)-Hydroxy Side Chain and p-Substituted Benzoate Function at C-23-Synthesis and Evaluation of Plant Growth Effects. Int. J. Mol. Sci. 2024, 25(14), 7515.
- Valdés, E.; Díaz, K.; Núñez, M.; Olea, A.F.; Moral, J.F.Q.d.; Carvajal, R.; Cuellar, M.A.; Espinoza-Catalán, L. Novel 3-Dehydroteasterone Derivatives with 23,24-Dinorcholanic Side Chain and Benzoate Groups at C-22: Synthesis and Activity Evaluation by Rice Lamina Inclination Test and Bean Second-Internode Bioassay. Int. J. Mol. Sci. 2025, 26(17), 8710.
- Carvajal, R.; Gonzalez, C.; Olea, A. F.; Fuentealba, M.; Espinoza, L. Synthesis of 2-Deoxybrassinosteroids Analogs with 24-nor, 22(S)-23-Dihydroxy-Type Side Chains from Hyodeoxycholic Acid. Molecules 2028, 23(6), 1306.
- Kvasnica, M.; Oklestkova, J.; Bazgier, V.; Rárová, L.; Korinkova, P.; Mikulík, J.; Budesinsky, M.; Béres, T.; Berka, K.; Lu, Q. Design, synthesis and biological activities of new brassinosteroid analogues with a phenyl group in the side chain. Org. Biomol. Chem. 2016, 14(37), 8691-8701.


