
- Organometallic-1,2,3-triazole,
- Cyrhetrene,
- Ferrocene,
- Aminomethyl,
- Reactivity
Copyright (c) 2026 SCHQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
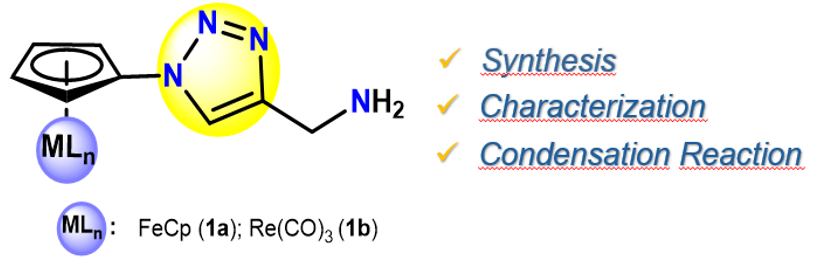
In search of new organometallic-1,2,3-triazoles, this work describes a convenient synthesis to obtain ferrocenyl and cyrhetrenyl 1,2,3-triazole derivatives containing aminomethyl fragment. On this regard, the compounds of general formulae [(η⁵-C₅H₄-(1)-1,2,3-triazole-(4)-CH₂NH₂)MLn] [where MLn =Fe(η⁵-C₅H₅) (1a), Re(CO)₃ (1b)] were obtained via tert-butoxycarbonyl (BOC) deprotection under acid conditions of the corresponding protected triazoles [(η⁵-C₅H₄-(1)-1,2,3-triazole-(4)-CH₂NH(C=O)OC(CH3)3)MLn] with MLn = Fe(η⁵-C₅H₅) (P1), Re(CO)₃ (P2)], with good yields (89–95%). In addition, the reactivity of aminomethyl compounds (1a–b) was evaluated in condensation reactions with 4-(1H-1,2,4-triazol-1-yl)benzaldehyde, isolated the Schiff bases [(η⁵-C₅H₄-(1)-1,2,3-triazole-(4)-CH₂N=CH-(1)-C₆H₄-(4)-1H-1,2,4-triazole)MLn] (2a–b) [where MLn =Fe(η⁵-C₅H₅) (2a), Re(CO)₃ (2b)] under mild reactions conditions. All compounds were characterized by FT-IR, ¹H NMR spectroscopy and elemental analysis. Moreover, the molecular structure of 2a was determined by single-crystal X-ray diffraction.
References
- C. Elschenbroich, Organometallics, 3rd ed., Wiley-VCH, (2006)
- R. H. Crabtree, The Organometallic Chemistry of the Transition Metals, 5th ed., John Wiley & Sons, (2009)
- S. D. Pike; A. S. Weller, Philos. Trans. R. Soc. A, 373, 20140187, (2015)
- P. Omer; E. Abdulkareem; R. Omer; R. Faruq Rashid, Rev. Inorg. Chem., 45, 397, (2025)
- G. Gasser; I. Ott; N. Metzler-Nolte, J. Med. Chem., 54, 3, (2011)
- K. D. Mjøs; C. Orvig, Chem. Rev., 114, 4540, (2014)
- M. Patra; G. Gasser, Nat. Rev. Chem., 1, 0066, (2017)
- C. G. Hartinger; N. Metzler-Nolte; P. J. Dyson, Organometallics, 31, 5677, (2012)
- A. Leonidova; G. Gasser, ACS Chem. Biol., 9, 2180, (2014)
- C. Ornelas; D. Astruc, Pharmaceutics, 15, 2044, (2023)
- T. Kealy; P. Pauson, Nature, 168, 1039, (1951)
- G. Wilkinson; M. Rosenblum; M. C. Whiting; R. B. Woodward, J. Am. Chem. Soc., 74, 2125, (1952)
- H. Werner, Angew. Chem. Int. Ed., 22, 927, (1983)
- J. Gómez; D. Sierra; M. Fuentealba; V. Artigas; A. H. Klahn, J. Organomet. Chem., 883, 65, (2019)
- R. Arancibia; A. H. Klahn; G. E. Buono-Core; D. Contreras; G. Barriga; C. Olea-Azar; M. Lapier; J. D. Maya; A. Ibañez; M. T. Garland, J. Organomet. Chem., 743, 49, (2013)
- C. Quintana; A. H. Klahn; V. Artigas; M. Fuentealba; C. Biot; I. Halloum; L. Kremer; R. Arancibia, Inorg. Chem. Commun., 55, 48, (2015)
- T. Maldonado; G. Ferraudi; A. G. Lappin; F. Godoy, J. Phys. Chem. A, 123, 9274, (2019)
- R. Arancibia; A. H. Klahn; M. Lapier; J. D. Maya; A. Ibáñez; M. T. Garland; S. Carrère-Kremer; L. Kremer; C. Biot, J. Organomet. Chem., 755, 1, (2014)
- V. V. Rostovtsev; L. G. Green; V. V. Fokin; K. B. Sharpless, Angew. Chem. Int. Ed., 41, 2596, (2002)
- L. Liang; D. Astruc, Coord. Chem. Rev., 255, 2933, (2011)
- V. Ganesh; V. S. Sudhir; T. Kundu; S. Chandrasekaran, Chem. Asian J., 6, 2670, (2011)
- B. Beyer; C. Ulbricht; D. Escudero; C. Friebe; A. Winter; L. González; U. S. Schubert, Organometallics, 28, 5478, (2009)
- F. Saleem; G. K. Rao; A. Kumar; G. Mukherjee; A. K. Singh, Organometallics, 32, 3595, (2013)
- D. P. Day; T. Dann; R. J. Blagg; G. G. Wildgoose, J. Organomet. Chem., 770, 29, (2014)
- P. Biegański; E. Kovalski; N. Israel; E. Dmitrieva; D. Trzybiński; K. Woźniak; V. Vrček; M. Godel; C. Riganti; J. Kopecka; H. Lang; K. Kowalski, Inorg. Chem., 61, 9650, (2022)
- D. Coelho; Y. Colas; M. Ethève-Quelquejeu; E. Braud; L. Iannazzo, ChemBioChem, 25, e202400150, (2024)
- J. Ceramella; D. Iacopetta; A. Catalano; F. Cirillo; R. Lappano; M. S. Sinicropi, Antibiotics (Basel)., 11, 191, (2022)
- R. Aggarwal; G. Sumran, Eur. J. Med. Chem., 205, 112652, (2020)
- O. Guerret; S. Solé; H. Gornitzka; G. Trinquier; G. Bertrand, J. Organomet. Chem., 600, 112, (2000)
- J. Liu; L. Li; H. Dai; Z. Liu; J. Fang, J. Organomet. Chem., 691, 2686, (2006)
- B. D. Sadanala; R. Trivedi, Chem. Rec., 24, e202300347, (2024)
- H. S. Scott; A. Nafady; J. D. Cashion; A. M. Bond; B. Moubaraki; K. S. Murray; S. M. Neville, Dalton Trans., 42, 10168, (2013)
- Z. Jin; A. Huo; T. Liu; Y. Hu; J. Liu; J. Fang, J. Organomet. Chem., 690, 1226, (2005)
- K. Yin; Y. Ruan; M. Guo; Q. Tang; F. Nie; Y. Wang; H. Guo; Y. Wang; S. Zhou; D. Yang; Y. Tang; R. Jin; H. Peng, Bioorg. Chem., 165, 108936, (2025)
- Manuscript submitted to J. Organomet. Chem., (submitted)
- W. L. F. Armarego; C. L. L. Chai, Purification of Laboratory Chemicals, 5th ed., Butterworth-Heinemann, (2003)
- Bruker. APEX5. Bruker AXS Inc., Madison, Wisconsin, USA, (2023)
- G. M. Sheldrick, SADABS, Program for Empirical Absorption Correction of Area Detector Data, (1996)
- G. M. Sheldrick, Acta Crystallogr., A71, 3, (2015)
- G. M. Sheldrick, Acta Crystallogr., C71, 3, (2015)
- O. V. Dolomanov; L. J. Bourhis; R. J. Gildea; J. A. K. Howard; H. Puschmann, J. Appl. Crystallogr., 42, 339, (2009)
- Z. Öztürk; Y. Yıldız; N. Abul; O. Ertik; F. Arı; İ. Gülçin; Ö. Koz; G. Koz, Polyhedron., 279, 117620, (2025)
- P. Toro; A. H. Klahn; B. Pradines; F. Lahoz; A. Pascual; C. Biot; R. Arancibia, Inorg. Chem. Commun., 35, 126, (2013)
- Y. Huentupil; L. Peña; N. Novoa; E. Berrino; R. Arancibia; C. T. Supuran, J. Enzyme Inhib. Med. Chem., 34, 451, (2019)
- C. Quintana; G. Silva; A. H. Klahn; V. Artigas; M. Fuentealba; C. Biot; I. Halloum; L. Kremer; N. Novoa; R. Arancibia, Polyhedron., 134, 166, (2017)
- F. Otón; M. del C. González; A. Espinosa; A. Tárraga; P. Molina, Organometallics, 31, 1758, (2012)
- K. Al Khalyfeh; A. Ghazzy; R. M. Al-As’ Ad; T. Rüffer; O. Kanoun; H. Lang, RSC Adv., 14, 20572, (2024)


