Comparative Ion Exchange Performance of Iminodiacetate and Aminophosphonate Resins for Efficient Cadmium Removal from Aqueous Solutions

- Cadmium removal,
- Chelating ion exchange resin,
- Iminodiacetic acid,
- Aminophosphonic acid,
- Regeneration
Copyright (c) 2026 SCHQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
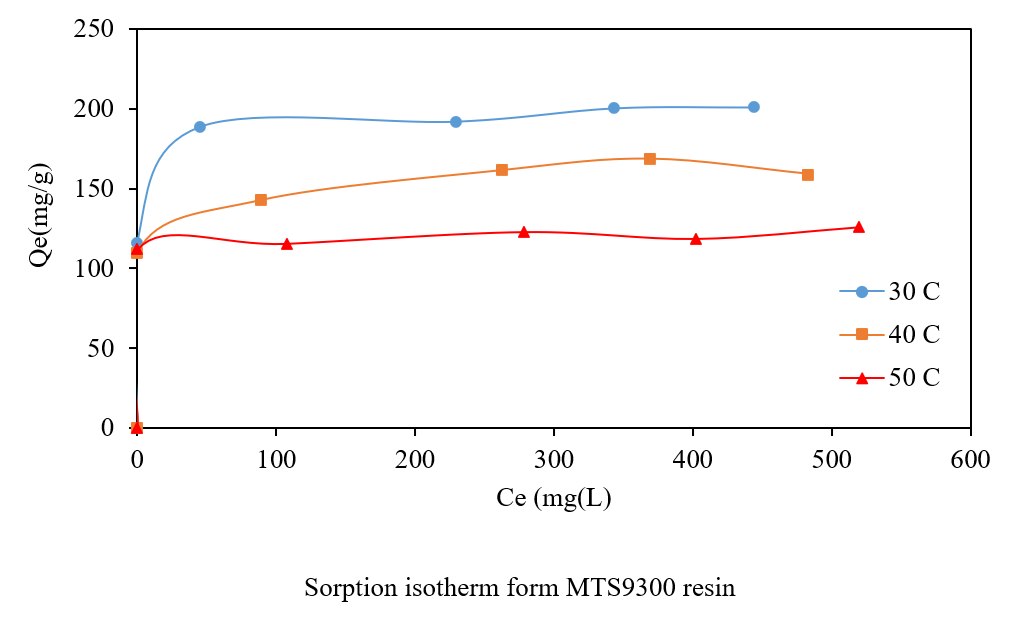
The removal of cadmium ions (Cd2+) from aqueous media was systematically investigated using two commercial chelating ion-exchange resins: Puromet MTS9300, functionalized with iminodiacetic acid groups, and Puromet MTS9500, containing aminophosphonic acid groups. The effects of resin dosage, solution pH, temperature, and contact time on Cd2+ removal were evaluated through batch experiments. The results revealed that both resins exhibited high Cd2+ removal efficiencies, achieving complete removal under optimized conditions. Ion exchange performance was strongly influenced by solution pH, as both resins possess weakly acidic functional groups that undergo protonation in acidic media, thereby reducing Cd2+ uptake. Kinetic data fitted well to the pseudo-second-order model. The equilibrium data correlated closely with the Langmuir model, with maximum exchange capacities of 201.33 mg/g for MTS9300 and 191.44 mg/g for MTS9500 at 30 °C. Thermodynamic analysis confirmed the spontaneous nature of Cd²⁺ removal for both resins, with ΔG° values ranging from –26.37 to –30.09 kJ/mol. MTS9500 exhibited an endothermic exchange process, while MTS9300 displayed exothermic behaviour. Regeneration experiments demonstrated excellent reusability with both HCl and H2SO4 solutions, maintaining nearly 100% regeneration efficiency. These findings highlight the high potential of MTS9300 and MTS9500 resins as efficient, regenerable materials for the removal of cadmium ions from contaminated water.
References
- R. Wang, P. Sang, Y. Guo, P. Jin, Y. Cheng, H. Yu, Y. Xie, W. Yao, and H. Qian, Cadmium in food: Source, distribution and removal, Food Chem. 405, 134666 (2023).
- M. M. Kwikima, S. Mateso, and Y. Chebude, Potentials of agricultural wastes as the ultimate alternative adsorbent for cadmium removal from wastewater. A review, Sci. African 13, e00934 (2021).
- B. Wang, J. Lan, J. Pu, X. Liu, and B. Gong, Adsorption of cadmium ions from simulated battery wastewater by polyethylene polyamine-modified activated carbon, Water Sci. Technol. 84, 3916 (2021).
- G. Genchi, M. S. Sinicropi, G. Lauria, A. Carocci, and A. Catalano, The Effects of Cadmium Toxicity, Int. J. Environ. Res. Public Health 17, 3782 (2020).
- C. Wang, H. Yin, L. Bi, J. Su, M. Zhang, T. Lyu, M. Cooper, and G. Pan, Highly efficient and irreversible removal of cadmium through the formation of a solid solution, J. Hazard. Mater. 384, 121461 (2020).
- F. Javaheri, Z. Kheshti, S. Ghasemi, and A. Altaee, Enhancement of Cd2+ removal from aqueous solution by multifunctional mesoporous silica: Equilibrium isotherms and kinetics study, Sep. Purif. Technol. 224, 199 (2019).
- K. Attar, D. Bouazza, H. Miloudi, A. Tayeb, A. Boos, A. M. Sastre, and H. Demey, Cadmium removal by a low-cost magadiite-based material: Characterization and sorption applications, J. Environ. Chem. Eng. 6, 5351 (2018).
- W. Zhang, Y. An, S. Li, Z. Liu, Z. Chen, Y. Ren, S. Wang, X. Zhang, and X. Wang, Enhanced heavy metal removal from an aqueous environment using an eco-friendly and sustainable adsorbent, Sci. Rep. 10, 16453 (2020).
- A. Reda, A.-G. El-Demerdash, W. Sadik, E. El-Rafey, and T. Shoeib, Effectively eliminating lead and cadmium from industrial wastewater using a biowaste-based sorbent, Appl. Water Sci. 15, 25 (2025).
- Z. Khan, A. Elahi, D. A. Bukhari, and A. Rehman, Cadmium sources, toxicity, resistance and removal by microorganisms-A potential strategy for cadmium eradication, J. Saudi Chem. Soc. 26, 101569 (2022).
- M. Khairy, S. A. El-Safty, and M. A. Shenashen, Environmental remediation and monitoring of cadmium, TrAC Trends Anal. Chem. 62, 56 (2014).
- N. H. A. Hamid et al., A state-of-art review on the sustainable technologies for cadmium removal from wastewater, Water Reuse 14, 312 (2024).
- K. Bhattacharyya, D. Sen, A. K. Banik, and S. Ganguly, Adsorptive removal of cadmium from aqueous medium-a critical review, Phys. Chem. Earth, Parts A/B/C 134, 103538 (2024).
- K. Pyrzynska, Removal of cadmium from wastewaters with low-cost adsorbents, J. Environ. Chem. Eng. 7, 102795 (2019).
- M. Kumar, A. Kushwaha, L. Goswami, A. K. Singh, and M. Sikandar, A review on advances and mechanism for the phycoremediation of cadmium contaminated wastewater, Clean. Eng. Technol. 5, 100288 (2021).
- A. E. D. Mahmoud, K. M. Al-Qahtani, S. O. Alflaij, S. F. Al-Qahtani, and F. A. Alsamhan, Green copper oxide nanoparticles for lead, nickel, and cadmium removal from contaminated water, Sci. Rep. 11, 1 (2021).
- B. Kayranli, Cadmium removal mechanisms from aqueous solution by using recycled lignocelluloses, Alexandria Eng. J. 61, 443 (2022).
- N. T. Abdel-Ghani, M. M. Hefny, G. A. El-Chaghaby, Removal of metal ions from synthetic wastewater by adsorption onto eucalyptus camaldulenis tree leaves, J. Chil. Chem. Soc. 53, 1585 (2008).
- N. M. Marin, M. Nita Lazar, M. Popa, T. Galaon, and L. F. Pascu, Current Trends in Development and Use of Polymeric Ion-Exchange Resins in Wastewater Treatment, Materials (Basel). 17, 5994 (2024).
- A. B. Botelho Junior, D. B. Dreisinger, and D. C. R. Espinosa, A Review of Nickel, Copper, and Cobalt Recovery by Chelating Ion Exchange Resins from Mining Processes and Mining Tailings, Mining, Metall. Explor. 36, 199 (2019).
- P. Barbaro and F. Liguori, Ion Exchange Resins: Catalyst Recovery and Recycle, Chem. Rev. 109, 515 (2009).
- S. Ghafoor, S. Ata, Synthesis of carboxyl-modified Fe3O4@SiO2 nanoparticles and their utilization for the remediation of cadmium and nickel from aqueous solution, J. Chil. Chem. Soc. 62, 3588 (2017).
- P. Rudnicki, Z. Hubicki, and D. Kołodyńska, Evaluation of heavy metal ions removal from acidic waste water streams, Chem. Eng. J. 252, 362 (2014).
- D. Kołodyńska, P. Rudnicki, and Z. Hubicki, New approach to Cu(II), Zn(II) and Ni(II) ions removal at high NaCl concentration on the modified chelating resin, Desalin. Water Treat. 74, 184 (2017).
- G. McKay, Y. S. Ho, and J. C. Y. Ng, Biosorption of Copper from Waste Waters: A Review, Sep. Purif. Rev. 28, 87 (1999).
- Y. . Ho and G. McKay, Pseudo-second order model for sorption processes, Process Biochem. 34, 451 (1999).
- M. Özacar and İ. A. Şengil, Adsorption of metal complex dyes from aqueous solutions by pine sawdust, Bioresour. Technol. 96, 791 (2005).
- B. Alyüz and S. Veli, Kinetics and equilibrium studies for the removal of nickel and zinc from aqueous solutions by ion exchange resins, J. Hazard. Mater. 167, 482 (2009).
- C. M. Simonescu, V. Lavric, A. Musina, O. M. Antonescu, D. C. Culita, V. Marinescu, C. Tardei, O. Oprea, and A. M. Pandele, Experimental and modeling of cadmium ions removal by chelating resins, J. Mol. Liq. 307, 112973 (2020).
- C.-W. Wong, J. P. Barford, G. Chen, and G. McKay, Kinetics and equilibrium studies for the removal of cadmium ions by ion exchange resin, J. Environ. Chem. Eng. 2, 698 (2014).
- D. Bilba, N. Bilba, and M. Albu, Kinetics of cadmium ion sorption on ion exchange and chelating resins, Solvent Extr. Ion Exch. 17, 1557 (1999).
- S. Elfeghe, S. Anwar, and Y. Zhang, Adsorption and removal studies of cadmium ion onto sulphonic/phosphonic acid functionalization resins, Can. J. Chem. Eng. 100, 3006 (2022).
- Y. Zhang, X. Duan, C. Hu, G. Du, and Y. Wang, Green and Cost-Effective Separation of Cadmium from Base Metals in Chloride Medium with Halide-Loaded Anion Exchanger, Processes 11, 1051 (2023).
- O. E. Roshdy, Removal of uranium, cadmium and iron ions from phosphoric acid solution using amberjet 1200 H resin: an experimental, isotherm and kinetic study, J. Radioanal. Nucl. Chem. 329, 85 (2021).
- A. Tunçeli, A. Ulaş, O. Acar, and A. R. Türker, Adsorption isotherms, kinetic and thermodynamic studies for cadmium and lead ions from water solutions using Amberlyst 15 resin, Turkish J. Chem. 46, 193 (2021).
- E. C. Lima, A. Hosseini-Bandegharaei, J. C. Moreno-Piraján, and I. Anastopoulos, A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t Hoof equation for calculation of thermodynamic parameters of adsorption, J. Mol. Liq. 273, 425 (2019).


