ANALYSIS OF CARILITE OLIGOMERS AND THEIR FUNCTIONALIZATION WITH FURFURYLAMINE AT LOW CONVERSION BY GPC, NMR AND ESI-MASS SPECTROSCOPIES

- Carilite oligomers,
- Paal-Knorr reaction,
- Diels-Alder,
- reversible crosslinking,
- polymer matrices
Copyright (c) 2026 SCHQ

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
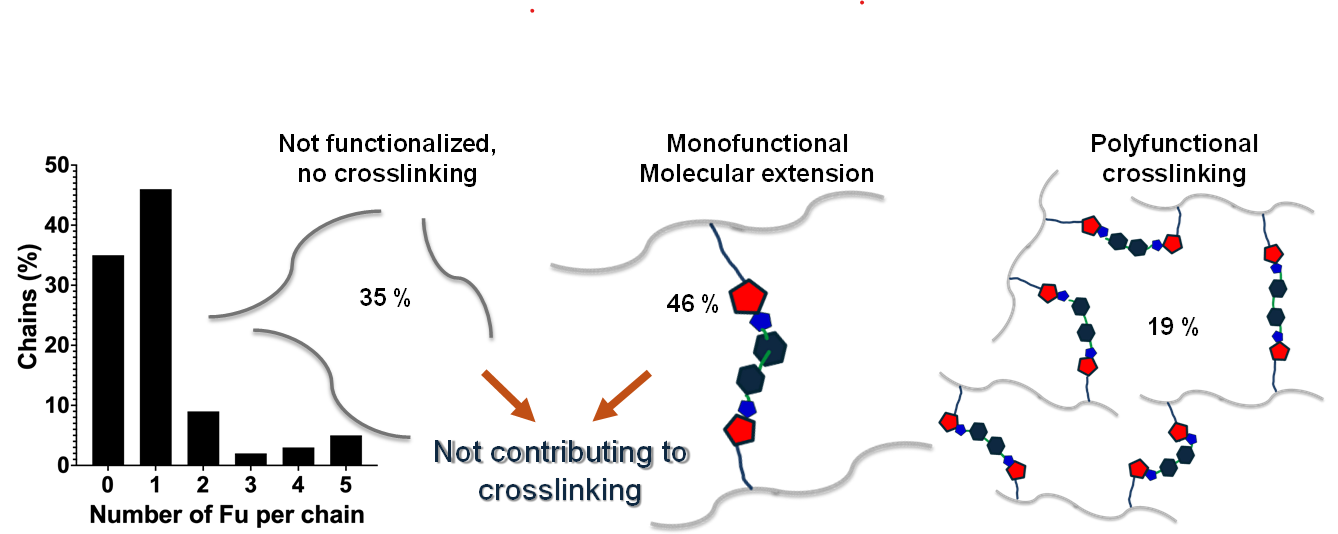
Background/Objectives: Carilite oligomers may be easily functionalized with primary amines, such as furfuryl amine, through the Paal-Knorr reaction, converting polymeric 1,4-diketone groups into N-substituted pyrroles with pendant furanyl groups. The resulting oligomers allow producing polymer and polymer composites materials by adding a bifunctional dienophile, leading to reversible crosslinking through the Diels-Alder reaction. The extent of conversion in the Paal Knorr reaction should determine the amount of polymer chains showing two or more furanyl dienes, condition necessary for extended crosslinking. Thus, the objectives of this work consisted of i) to characterize a sample of Carilite oligomers (PK30), ii) to calculate the probabilistic distribution of the N-substituted pyrroles in the oligomer chains as a function of the diketone conversion, and iii) to compare the prediction with experimental data of a sample obtained after reaction between PK30 and furfuryl amine aiming at 20 % of conversion. Methods: GPC, NMR, and ESI mass spectroscopies are used for the analysis of the polymers. Poisson distribution and one-dimensional hard-dimer exclusion models have been applied for probabilistic calculations. Results: Carilite oligomers are a polydisperse mixture where the most abundant molecules present 1 – 16 ketone moieties. After functionalization, the most abundant derivatized molecules consist of oligomers functionalized with only one furfuryl amino residue. Good matching with the predictions were found. Conclusions: The use of NMR combined with ESI-mass spectroscopy has served to understand the molecular structure of Carilite oligomers and their furfuryl amine-functionalized derivatives. This allows determining refined molecular weights that allow calculating effective conversion aimed and, in combination with the probabilistic predictions, obtaining insights of the expected Fu content per chain, contributing to the improvement of design, handling, and control strategies for reversibly crosslinked polymer matrices.
References
- (1) Zhu, L.; Li, J.; Chen, W.-Y.; Ziegler, C. J.; Takahashi, K.; Nozaki, K.; Jia, L. Aliphatic Polyketones from Alternating Copolymerization of CO and Olefins: Phosphinoamidate Nickel Catalyst, Polymerization Study, Mechanical Properties and Degradations. ACS Appl. Polym. Mater. 2024, 6 (16), 9829–9836. https://doi.org/10.1021/acsapm.4c01691.
- (2) Mul, W. P.; Dirkzwager, H.; Broekhuis, A. A.; Heeres, H. J.; Van Der Linden, A. J.; Guy Orpen, A. Highly Active, Recyclable Catalyst for the Manufacture of Viscous, Low Molecular Weight, CO–Ethene–Propene-Based Polyketone, Base Component for a New Class of Resins. Inorgani-ca Chim. Acta 2002, 327 (1), 147–159. https://doi.org/10.1016/S0020-1693(01)00697-1.
- (3) González Cortes, P.; Araya-Hermosilla, R.; Wrighton-Araneda, K.; Cortés-Arriagada, D.; Pic-chioni, F.; Yan, F.; Rudolf, P.; Bose, R. K.; Quero, F. Effect of Intermolecular Interactions on the Glass Transition Temperature of Chemically Modified Alternating Polyketones. Mater. To-day Chem. 2023, 34, 101771. https://doi.org/10.1016/j.mtchem.2023.101771.
- (4) Toncelli, C.; Schoonhoven, M.-J.; Broekhuis, A. A.; Picchioni, F. Paal-Knorr Kinetics in Wa-terborne Polyketone-Based Formulations as Modulating Cross-Linking Tool in Electrodeposi-tion Coatings. Mater. Des. 2016, 108, 718–724. https://doi.org/10.1016/j.matdes.2016.06.127.
- (5) Paal‐Knorr Pyrrole Synthesis. In Comprehensive Organic Name Reactions and Reagents; Wiley, 2010; pp 2107–2110. https://doi.org/10.1002/9780470638859.conrr475.
- (6) Araya-Hermosilla, R.; Lima, G. M. R.; Raffa, P.; Fortunato, G.; Pucci, A.; Flores, M. E.; More-no-Villoslada, I.; Broekhuis, A. A.; Picchioni, F. Intrinsic Self-Healing Thermoset through Co-valent and Hydrogen Bonding Interactions. Eur. Polym. J. 2016, 81, 186–197. https://doi.org/10.1016/j.eurpolymj.2016.06.004.
- (7) Araya-Hermosilla, E.; Roscam Abbing, M.; Catalán-Toledo, J.; Oyarzun-Ampuero, F.; Pucci, A.; Raffa, P.; Picchioni, F.; Moreno-Villoslada, I. Synthesis of Tuneable Amphiphilic-Modified Polyketone Polymers, Their Complexes with 5,10,15,20-Tetrakis-(4-Sulfonatophenyl)Porphyrin, and Their Role in the Photooxidation of 1,3,5-Triphenylformazan Confined in Polymeric Na-noparticles. Polymer 2019, 167, 215–223. https://doi.org/10.1016/j.polymer.2019.01.079.
- (8) Orozco, F.; Li, J.; Ezekiel, U.; Niyazov, Z.; Floyd, L.; Lima, G. M. R.; Winkelman, J. G. M.; Moreno-Villoslada, I.; Picchioni, F.; Bose, R. K. Diels-Alder-Based Thermo-Reversibly Cross-linked Polymers: Interplay of Crosslinking Density, Network Mobility, Kinetics and Stereoi-somerism. Eur. Polym. J. 2020, 135, 109882. https://doi.org/10.1016/j.eurpolymj.2020.109882.
- (9) Orozco, F.; Kaveh, M.; Santosa, D. S.; Lima, G. M. R.; Gomes, D. R.; Pei, Y.; Araya-Hermosilla, R.; Moreno-Villoslada, I.; Picchioni, F.; Bose, R. K. Electroactive Self-Healing Shape Memory Polymer Composites Based on Diels–Alder Chemistry. ACS Appl. Polym. Mater. 2021, 3 (12), 6147–6156. https://doi.org/10.1021/acsapm.1c00999.
- (10) Araya-Hermosilla, E.; Carlotti, M.; Orozco, F.; Lima, G. M. R.; Araya-Hermosilla, R.; Ortega, D. E.; Cortés-Arriagada, D.; Picchioni, F.; Bose, R. K.; Mattoli, V.; Pucci, A. Tailoring Ther-momechanical, Shape Memory and Self-Healing Properties of Furan-Based Polyketone via Diels-Alder Chemistry with Different Bismaleimide Crosslinkers. Polymers 2025, 17 (5), 565. https://doi.org/10.3390/polym17050565.
- (11) Zhang, W.-L.; Han, S.; Li, S.-H.; Hao, X.-Y.; Lu, X.-B.; Liu, Y. Mechanistic Insights into Cat-ionic [P,O]-Pd-Catalyzed Chain-Transfer Copolymerization of Ethylene with Carbon Monoxide. Macromolecules 2024, 57 (9), 4174–4183. https://doi.org/10.1021/acs.macromol.4c00094.
- (12) Gooranorimi, A.; Mousavifard, S. M.; Mohseni, M.; Yahyaei, H.; Makki, H. Effective Cross-Link Density as a Metric for Structure–Property Relationships in Complex Polymer Net-works: Insights from Acrylic Melamine Systems. ACS Appl. Polym. Mater. 2025, 7 (14), 9034–9044. https://doi.org/10.1021/acsapm.5c01155.
- (13) Torres-Knoop, A.; Schamboeck, V.; Govindarajan, N.; Iedema, P. D.; Kryven, I. Effect of Dif-ferent Monomer Precursors with Identical Functionality on the Properties of the Polymer Net-work. Commun. Mater. 2021, 2 (1), 50. https://doi.org/10.1038/s43246-021-00154-x.
- (14) Malinova, V.; Rieger, B. Synthesis of Functional Poly(1,4-Ketone)s Bearing Bioactive Moie-ties by Pd-Catalyzed Insertion Polymerization. Biomacromolecules 2006, 7 (11), 2931–2936. https://doi.org/10.1021/bm0606364.
- (15) Zhang, Y.; Broekhuis, A. A.; Picchioni, F. Thermally Self-Healing Polymeric Materials: The Next Step to Recycling Thermoset Polymers? Macromolecules 2009, 42 (6), 1906–1912. https://doi.org/10.1021/ma8027672.
- (16) Drent, E.; Budzelaar, P. H. M. Palladium-Catalyzed Alternating Copolymerization of Alkenes and Carbon Monoxide. Chem. Rev. 1996, 96 (2), 663–682. https://doi.org/10.1021/cr940282j.
- (17) Gody, G.; Zetterlund, P. B.; Perrier, S.; Harrisson, S. The Limits of Precision Monomer Place-ment in Chain Growth Polymerization. Nat. Commun. 2016, 7 (1), 10514. https://doi.org/10.1038/ncomms10514.
- (18) Izunobi, J. U.; Higginbotham, C. L. Polymer Molecular Weight Analysis by1 H NMR Spec-troscopy. J. Chem. Educ. 2011, 88 (8), 1098–1104. https://doi.org/10.1021/ed100461v.
- (19) Siddiqui, M. N.; Ali, M. F. Investigation of Chemical Transformations by NMR and GPC dur-ing the Laboratory Aging of Arabian Asphalt. Fuel 1999, 78 (12), 1407–1416. https://doi.org/10.1016/S0016-2361(99)00080-0.
- (20) Bruns, H.; Ziesche, L.; Taniwal, N. K.; Wolter, L.; Brinkhoff, T.; Herrmann, J.; Müller, R.; Schulz, S. N -Acylated Amino Acid Methyl Esters from Marine Roseobacter Group Bacteria. Beilstein J. Org. Chem. 2018, 14, 2964–2973. https://doi.org/10.3762/bjoc.14.276.
- (21) Demarque, D. P.; Crotti, A. E. M.; Vessecchi, R.; Lopes, J. L. C.; Lopes, N. P. Fragmentation Reactions Using Electrospray Ionization Mass Spectrometry: An Important Tool for the Struc-tural Elucidation and Characterization of Synthetic and Natural Products. Nat. Prod. Rep. 2016, 33 (3), 432–455. https://doi.org/10.1039/C5NP00073D.
- (22) Grossert, J. S.; Fancy, P. D.; White, R. L. Fragmentation Pathways of Negative Ions Produced by Electrospray Ionization of Acyclic Dicarboxylic Acids and Derivatives. Can. J. Chem. 2005, 83 (11), 1878–1890. https://doi.org/10.1139/v05-214.
- (23) Mutenda, K. E.; Körner, R.; Christensen, T. M. I. E.; Mikkelsen, J.; Roepstorff, P. Application of Mass Spectrometry to Determine the Activity and Specificity of Pectin Lyase A. Carbohydr. Res. 2002, 337 (13), 1217–1227. https://doi.org/10.1016/S0008-6215(02)00127-1.

