DONNAN DIALYSIS ASSISTED BY INTERPENETRATING POLYMER NETWORKS FOR CHROMIUM ION TRANSPORT IN AQUEOUS MEDIA
- Chromium,
- Donnan dialysis,
- interpenetrating polymer network,
- mathematical model,
- polypropylene
Copyright (c) 2018 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
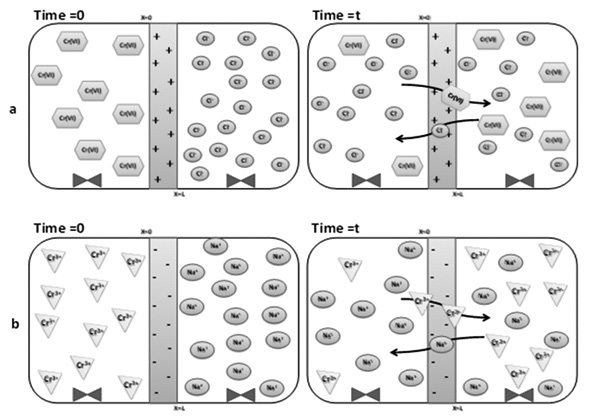
Macroporous polypropylene (MPP) membranes incorporating poly[sodium (styrene sulphonate)], P(SSNa), and poly[(ar-vinylbenzyl) trimethylammonium chloride],P(ClVBTA) were modified via “in situ” radical polymerization. Modified and unmodified MPP were characterized using SEM and charge density properties. Donnan dialysis was used to transport chromium ions. The results show that the degree of modification varied between 2.5% and 4.0%, and the water uptake percentage varied between 15% and 20%. Experimental data for chromium ion transport (Cr(III) and Cr(VI)) were fitted to a mathematical model, and a correlation coefficient very close to 1 was obtained. The main parameters of this mathematical fit are k and a. These parameters report the concentration of chromium ions that converge in the Donnan equilibrium (k) and the response rate of the modified membrane (a).
References
- S. Ballesteros, J. M Rincón, B. Rincón-Mora, M. Jordán, Vitrification of urban soil contamination by hexavalent chromium, J. Geochem. Explor. 2016.
- WHO, Guidelines for Drinking Water Quality, in, World Health Organization, Genova, Italy, 2008.
- W. Jin, H. Du, S. Zheng, Y. Zhang, Electrochemical processes for the environmental remediation of toxic Cr (VI): A review, Electrochim. Acta. 191, 1044, (2016).
- R. B. García-Reyes, J. R. Rangel-Mendez, M. C. Alfaro-De la Torre, Chromium (III) uptake by agro-waste biosorbents: Chemical characterization, sorption–desorption studies, and mechanism, J. Hazard. Mater. 170, 845, (2009).
- A. Golder, A. Samanta, S. Ray, Removal of trivalent chromium by electrocoagulation, Sep. Purif. Technol. 53, 33, (2007).
- R. R Patterson, S. Fendorf, M. Fendorf, Reduction of hexavalent chromium by amorphous iron sulfide, Environ. Sci Technol. 31, 2039, (1997).
- D. Park, S.-R. Lim, Y.-S. Yun, J.M. Park, Reliable evidences that the removal mechanism of hexavalent chromium by natural biomaterials is adsorption-coupled reduction, Chemosphere. 70, 298, (2007).
- S. A. Cavaco, S. Fernandes, M. M. Quina, L. M. Ferreira, Removal of chromium from electroplating industry effluents by ion exchange resins, J. Hazard. Mater. 144, 634, (2007).
- A. Figoli, J. Hoinkis, J. Bundschuh, Membrane Technologies for Water Treatment: Removal of Toxic Trace Elements with Emphasis on Arsenic, Fluoride and Uranium, CRC Press. London, UK, 2016.
- M. San Román, E. Bringas, R. Ibanez, I. Ortiz, Liquid membrane technology: fundamentals and review of its applications, J. Chem. Technol. Biot. 85, 2, (2010).
- E. H Cwirko, R. G. Carbonell, A theoretical analysis of Donnan dialysis across charged porous membranes, J. Membr. Sci. 48, 155, (1990).
- K. Pyrzynska, Preconcentration and recovery of metal ions by Donnan dialysis, Microchim Acta. 153, 117, (2006).
- A. Tor, Y. Çengeloğlu, M. Ersöv, G. Arslan, Transport of chromium through cation-exchange membranesby Donnan dialysis in the presence of some metals of different valences, Desalination. 170, 151, (2004).
- M. J. Hatch, J. A. Dillon, Acid retardation. Simple physical method for separation of strong acids from their salts, Ind. Eng. Chem. Process Des. Dev. 2, 253, (1963).
- J. Mathur, M. Murali, M.B. Krishna, V. Ramachandhran, M. Hanra, B. Misra, Diffusion dialysis aided electrodialysis process for concentration of radionuclides in acid medium, J. Radioanal. Nucl. Chem. 232, 237, (1998).
- A. A. Said, M. Amara, H. Kerdjoudj, The effect of thiourea as a complexing agent on the separation of metallic ions through cation exchange membranes by Donnan dialysis, Ionics. 19, 177, (2013).
- S. Velizarov, J. G. Crespo, M. A. Reis, Removal of inorganic anions from drinking water supplies by membrane bio/processes, Rev. Environ. Sci. Bio/Technol. 3, 361, (2004).
- L. Chikh, V. Delhorbe, O. Fichet, (Semi-) Interpenetrating polymer networks as fuel cell membranes, J. Membr. Sci. 368, 1, (2011).
- N. Sahiner, W. Godbey, G. L. McPherson, V. T. John, Microgel, nanogel and hydrogel–hydrogel semi-IPN composites for biomedical applications: synthesis and characterization, Colloid Polym. Sci. 284, 1121, (2006).
- A. Bajpai, J. Bajpai, S. Shukla, Water sorption through a semi-interpenetrating polymer network (IPN) with hydrophilic and hydrophobic chains, React. Funct. Polym. 50, 9, (2002).
- B. Krajewska, Application of chitin-and chitosan-based materials for enzyme immobilizations: a review, Enzyme Microb. Technol. 35, 126, (2004).
- C. O. M’Bareck, Q. T. Nguyen, S. Alexandre, I. Zimmerlin, Fabrication of ion-exchange ultrafiltration membranes for water treatment: I. Semi-interpenetrating polymer networks of polysulfone and poly (acrylic acid), J. Membr. Sci. 278, 10, (2006).
- G. Couture, A. Alaaeddine, F. Boschet, B. Ameduri, Polymeric materials as anion-exchange membranes for alkaline fuel cells, Prog. Polym. Sci. 36, 1521, (2011).
- Y. S. Dzyazko, S. L. Vasilyuk, L. M. Rozhdestvenskaya, V. N. Belyakov, N. V. Stefanyak, N. Kabay, M. Yüksel, Ö. Arar, Ü. Yüksel, Electro-deionization of Cr (VI)-Containing Solution. Part II: Chromium transport through inorganic ion-exchanger and composite ceramic membrane, Chem. Eng. Commun. 196, 22, (2008).
- J. Sánchez, B. L. Rivas, Liquid‐Phase Polymer‐Based Retention of Chromate and Arsenate Oxy‐Anions, in: Macromol. Symp. Wiley Online Library, 2012; pp. 123-136.
- Y. Tapiero, B. L. Rivas, J. Sánchez, M. Bryjak, N. Kabay, Polypropylene membranes modified with interpenetrating polymer networks for the removal of chromium ions, J. Appl. Polym. Sci. 132, 41953, (2015).
- B. L. Rivas, E. D. Pereira, M. Palencia, J. Sánchez, Water-soluble functional polymers in conjunction with membranes to remove pollutant ions from aqueous solutions, Prog. Polym. Sci. 36, 294, (2011).
- J. Neter, M. H. Kutner, C. J. Nachtsheim, W. Wasserman, Applied linear statistical models, Irwin, Chicago, 1996.
- W. M. Deen, Analysis of transport phenomena (topics in chemical engineering),Oxford University Press, New York, USA, 1998.
- M. Bryjak, I. Duraj, Anion-exchange membranes for separation of borates by Donnan dialysis, Desalination. 310, 39, (2013).
- O. Thomas, C. Burgess, UV-visible Spectrophotometry of Water and Wastewater, Elsevier. , Amsterdam, Netherlands, 2007.
- A. Szymczyk, P. Fievet, J. Reggiani, J. Pagetti, Electrokinetic characterization of mixed alumina-titania-silica MF membranes by streaming potential measurements, Desalination. 115, 129, (1998).
- H. Matsuyama, M. Yuasa, Y. Kitamura, M. Teramoto, D.R. Lloyd, Structure control of anisotropic and asymmetric polypropylene membrane prepared by thermally induced phase separation, J. Membr. Sci. 179, 91, (2000).
- C. C. Wang, F.L. Yang, L. F. Liu, Z. M. Fu, Y. Xue, Hydrophilic and antibacterial properties of polyvinyl alcohol/4-vinylpyridine graft polymer modified polypropylene non-woven fabric membranes, J. Membr. Sci. 345, 223, (2009).
- Q. Yang, Z. -K. Xu, Z. -W. Dai, J. -L. Wang, M. Ulbricht, Surface modification of polypropylene microporous membranes with a novel glycopolymer, Chem. Mater. 17, 3050, (2005).
- J. Schauer, J. Hnát, L. Brožová, J. Žitka, K. Bouzek, Heterogeneous anion-selective membranes: Influence of a water-soluble component in the membrane on the morphology and ionic conductivity, J. Membr. Sci. 401, 83, (2012).
- T. Maddanimath, I. S. Mulla, S. R. Sainkar, K. Vijayamohanan, K. I. Shaikh, A. S. Patil, S. P. Vernekar, Humidity sensing properties of surface functionalized polyethylene and polypropylene films, Sens. Actuators, B. 81, 141, (2002).
- A. Szymczyk, P. Fievet, J. Reggiani, J. Pagetti, Characterisation of surface properties of ceramic membranes by streaming and membrane potentials, J. Membr. Sci. 146, 277, (1998).
- K. Hu, J. M. Dickson, Modelling of the pore structure variation with pH for pore-filled pH-sensitive poly (vinylidene fluoride)–poly (acrylic acid) membranes, J. Membr. Sci. 321, 162, (2008).
- H. -Y. Yu, Z. -K. Xu, Q. Yang, M. -X. Hu, S. -Y. Wang, Improvement of the antifouling characteristics for polypropylene microporous membranes by the sequential photoinduced graft polymerization of acrylic acid, J. Membr. Sci. 281, 658, (2006).
- D. H. Kim, S. -H. Moon, J. Cho, Investigation of the adsorption and transport of natural organic matter (NOM) in ion-exchange membranes, Desalination. 151, 11, (2003).
- Y. Tapiero, B. L. Rivas, J. Sánchez, Functional ion membranes supported inside microporous polypropylene membranes to transport chromium ions: determination of mass transport coefficient, J. Chil. Chem. Soc. 59, 2737, (2014).
- C. Fontàs, I. Queralt, M. Hidalgo, Novel and selective procedure for Cr (VI) determination by X-ray fluorescence analysis after membrane concentration, Spectrochim. Acta, part B. 61, 407, (2006).
- W. G. Cook, R. P. Olive, Pourbaix diagrams for chromium, aluminum and titanium extended to high-subcritical and low-supercritical conditions, Corros. Sci. 58, 291, (2012).
- M. Owlad, M. K. Aroua, W. A. W. Daud, S. Baroutian, Removal of hexavalent chromium-contaminated water and wastewater: a review, Water, Air, Soil Poll. 200, 59, (2009).
- J. Kotaś, Z. Stasicka, Chromium occurrence in the environment and methods of its speciation, Environ. Pollut. 107, 263, (2000).
- Y. Marcus, Thermodynamics of solvation of ions. Part 5.—Gibbs free energy of hydration at 298.15 K, J. Chem. Soc., Faraday Trans. 87, 2995, (1991).


