SIMULTANEOUS DETERMINATION OF SACCHARINE, CAFFEINE, SALICYLIC ACID AND BENZOIC ACID IN DIFFERENT MATRIXES BY SALT AND AIR-ASSISTED HOMOGENEOUS LIQUID-LIQUID EXTRACTION AND HIGH-PERFORMANCE LIQUID CHROMATOGRAPHY
- Homogeneous liquid-liquid extraction,
- Salt and air-assisted,
- Saccharine,
- Caffeine,
- Salicylic acid
- Benzoic acid,
- High-performance liquid chromatography ...More
Copyright (c) 2017 Rouhollah Heydari, Masoumeh Mousav

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
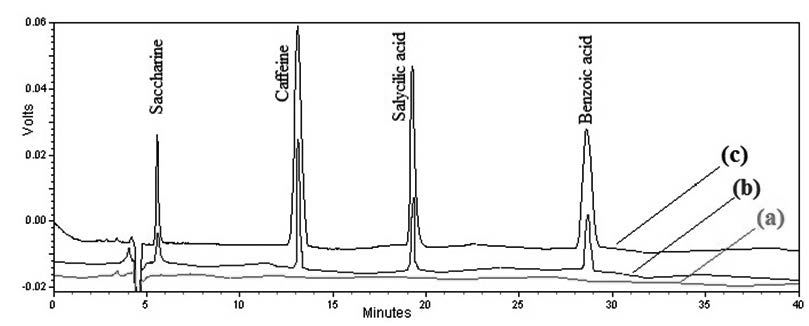
Saccharine, caffeine, salicylic acid and benzoic acid were extracted and quantified simultaneously by coupling the salt and air-assisted homogeneous liquid-liquid extraction (SAAHLLE) technique with high-performance liquid chromatography (HPLC). Various parameters such as extraction solvent and its volume, salt and its concentration, volume of injected air, vortex time and sample pH were evaluated and optimized. Analytical parameters of the proposed method were determined under the optimized conditions. The calibration curves showed good linearity in the range of 5-100 μg/mL for saccharine, caffeine, salicylic acid and benzoic acid. The limit of detection (LOD) values for saccharine, caffeine, salicylic acid and benzoic acid were 0.024, 0.013, 0.0048 and 0.0052 ng/mL, respectively. The recoveries were in the range of 88.0–94.0% with RSD values ranging from 3.8 to 6.8%.
References
- R. Rezaeepour, R. Heydari, A. Ismaili, Anal. Methods. 7, 3253, (2015).
- R. S. Razmara, A. Daneshfar, R. Sahrai, J. Ind. Eng. Chem. 17, 533 (2011).
- F. J. Zhao, H. Tang, Q. H. Zhang, J. Yang, A. K. Davey, J. P. Wang, J. Chromatogr. B. 881–882, 119, (2012).
- H. M. Tarkan, J. A. Finch, Miner. Eng. 18, 83, (2005).
- H. M. Tarkan, J. A. Finch, Colloid Sur. A. 264, 126, (2005).
- P. C. Lee, C. W. Li, S. S. Chen, C. H. Chiu, Separ. Sci. Technol. 44, 3911, (2009).
- C. W. Li, Y. M. Chen, S. T. Hsiao, Chemosphere. 71, 51, (2008).
- M. Hosseini, R. Heydari, M. Alimoradi, Talanta. 130, 171, (2014).
- R. Heydari, S. Zarabi, Anal. Methods. 6, 8469, (2014).
- A. O. Santini, S. C. Lemos, H. R. Pezza, J. Carloni-Filho, L. Pezza, Microchem. J. 90, 124, (2008).
- F. Han, Y. Z. He, L. Li, G. N. Fu, H. Y. Xie, W. E. Gan, Anal. Chim. Acta. 618, 79, (2008).
- X. Zhang, S. Xu, Y. Sun, Y. Wang, C. Wang, Chromatographia. 73, 1217, (2011).
- A. B. Bergamo, J. A. Fracassi da Silva, D. P. de Jesus, Food Chem. 124, 1714, (2011).
- E. Rudolph, A. Färbinger, J. König, Food Addit. Contam. A. 29, 1849, (2012).
- Z. Xia, Y. Ni, S. Kokot, Food Chem. 141, 4087, (2013).
- Y. Chen, H. Luo, X. Guo, Y. Xian, D. Luo, Y. Wu, Anal. Methods. 6, 4803, (2014).
- M. Grembecka, P. Baran, A. Blazewicz, Z. Fijalek, P. Szefer, Eur. Food Res. Technol. 238, 357, (2014).
- M. M. Karim, S. M. Alam, S. H. Lee, Luminescence. 23, 417, (2008).
- H. Parham, N. Rahbar, J. Pharm. Biomed. Anal. 50, 58, (2009).
- M. R. Payán, M. A. Bello López, R. Fernández-Torres, J. L. Pérez Bernal, M. C. Mochón, Anal. Chim. Acta. 653, 184, (2009).
- J. A. Murillo Pulgarín, A. A. Molina, I. S. F. Robles, Spectrochim. Acta A. 79, 909, (2011).
- R. M. Youssef, M. A. Korany, M. A. Afify, Anal. Methods. 6, 3410, (2014).
- T. Li, L. Zhang, L. Tong, Q. Liao, Biomed. Chromatogr. 28, 648, (2014).
- A. Koltsakidou, C. K. Zacharis, K. Fytianos, J. Chromatogr. A. 1377, 46, (2015).
- C. E. Matkovich, Anal. Chem. 45, 1915, (1973).
- T. Zhigang, Z. Rongqi, D. Zhanting, J. Chem. Technol. Biotechnol. 76, 757, (2001).
- P. D. Tzanavaras, D. G. Themelis, Anal. Chim. Acta. 581, 89, (2007).
- C. B. Mendes, E. P. Laignier, M. R. P. L. Brigagão, P. O. Luccas, C. R. T. Tarley, Chem. Pap. 64, 285, (2010).
- P. J. Tseng, C. Y. Wang, T. Y. Huang, Y. Y. Chuang, S. F. Fub, Y. W. Lin, Anal. Methods. 6, 1759, (2014).


