EFFECT OF CHEMICAL AND PHYSICAL VARIABLES IN THE PHOTO-ELECTROCHEMICAL REMOVAL OF ESTRIOL (E3) AND 17 α-ETHINYLESTRADIOL (EE2) IN AQUEOUS SOLUTION
- Photoelectroxidationn,
- Estriol,
- Ethynilestradiol,
- endocrine disruptor,
- DSA (Ti/RUO2)
Copyright (c) 2019 Journal of the Chilean Chemical Society

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Abstract
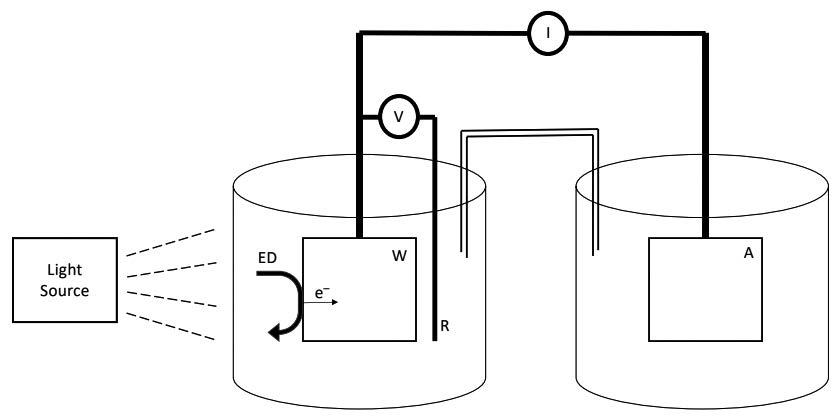
The effect of different variables in the electrochemical removal of estriol (E3) in aqueous solution was studied and it was compared with the removal of 17 α- Ethinylestradiol. The photoelectroxidations were carried out with DSA electrode (Ti/RuO2), varying the pH, substrate concentration and supporting electrolyte, potential configuration of saline bridge, reactor volume, and light source. Results for both Estriol and 17 α- Ethinylestradiol are similar, showing to be the best removals at concentrations of 500 ug/L of substrate, volumes to be treated of 90 ml of solution, saline bridge of 30 cm in length, voltages between 0.8 and 3, concentration of Na2SO4 of 0.1 M, pH between 6 and10, and UV lamp of 125 W.
References
- L.G. Parks, C.S. Lambright, E.F. Orlando, L.J. Guillette, G.T. Ankley, L.E. Gray. Masculinization of female mosquitofish in kraft mill effluent-contaminated Fenholloway River water is associated with androgen receptor agonist activity. Toxicological Sciences. (2001) 62 (2), pp. 257– 267.
- L. Clouzot, M. Benoît, D. Pierre, N. Roche. (2008). 17α‐Ethinylestradiol: An endocrine disrupter of great concern. Analytical methods and removal processes applied to water purification. A review. Environmental Progress & Sustainable Energy. (2008) 27 (3), pp. 383–396.
- R. J. Kavlock, G. P. Daston, C. De Rosa, P. Fenner-Crisp, L. E. Gray, S. Kaattari, G. Lucier, M. Luster, M. J. Mac, C. Maczka. Research needs for the risk assessment of health and environmental effects of endocrine disruptors: A report of the U.S. Environ. Health Perspect. (1996) 104 (Suppl. 4), pp. 715–740.
- C.P. Huang, C. Dong, Z Tang. Advanced chemical oxidation: its present role and potential future in hazardous waste treatment. Waste management. (1993) 13, pp. 361–377.
- X. Domenech, W. F. Jardim, M. I. Litter. “Eliminación de contaminantes por fotocatálisis heterogénea”, Miguel A. Blesa (Ed.), Procesos avanzados de oxidación para la eliminación de contaminantes, Red Cydted VIII, Buenos Aires, (2001), pp. 3-26.
- M. Klavarioti, D. Mantzavinos, D. Kassinos. Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environment international. (2009). 35 (2), pp. 402-417.
- Z.H. Liu, Y. Kanjo, S. Mizutani. Removal mechanisms for endocrine disrupting compounds (EDCs) in wastewater treatment—physical means, biodegradation, and chemical advanced oxidation: a review. Science of the Total Environment. (2009), 407 (2), pp. 731-748.
- A.C. Johnson, R. Williams. A model to estimate influent and effluent concentrations of estradiol, estrone, and ethinylestradiol at sewage treatment works. Environmental science & technology. (2004) 38 (13), pp. 3649-3658.
- A.Z. Aris, A. S. Shamsuddin, S.M. Praveena. Occurrence of 17α-ethynylestradiol (EE2) in the environment and effect on exposed biota: a review. Environment international. (2014) 69, pp. 104-119.
- H.M. Coleman, M.I. Abdullah, B.R. Eggins, F.L. Palmer. Photocatalytic degradation of 17β-oestradiol, oestriol and 17α-ethynyloestradiol in water monitored using fluorescence spectroscopy. Applied Catalysis B: Environmental. (2005) 55 (1), pp. 23-30.
- B. Cédat, C. De Brauer, H. Métivier, N. Dumont, R. Tutundjan. (2016). Are UV photolysis and UV/H2O2 process efficient to treat estrogens in waters Chemical and biological assessment at pilot scale. Water Research. (2016) 100 (1), pp.357-366.
- L.A. Racz, R.K. Goel. Fate and removal of estrogens in municipal wastewater. Journal of Environmental Monitoring. (2010), 12 (1), pp. 58- 70.
- M.F. Brugnera, K. Rajeshwar, J. Cardoso, M.V.B. Zanoni. Bisphenol A removal from wastewater using self-organized TIO 2 nanotubular array electrodes. Chemosphere. (2010) 78 (5), 569-575.
- C. Salazar, C.A. Zaror; M.A. M.A. Mondaca, P. Gonzalez; H. Mansilla, C. Pena. Advanced Electrochemical Oxidation of Ultrafiltration Permeates from Cellulose Bleaching Effluents. Journal of Advanced Oxidation Technologies. (2012), 15 (2) pp. 254-265.
- C.A. Zaror; C. Salazar, E.A. Araneda, M.A. Mondaca, H.D. Mansilla, C. Pena. Electrochemical Treatment of Segregated Effluents from the D-Stage in ECF Kraft Cellulose Bleaching. Journal of Advanced Oxidation Technologies. (2011), 14 (1), pp. 47-53.
- M.V.B. Zanoni, J.J. Sene, M.A. Anderson. Photoelectrocatalytic degradation of Remazol Brilliant Orange 3R on titanium dioxide thin-film electrodes. Journal of Photochemistry and Photobiology A: chemistry. (2003), 157 (1), pp. 55–63.
- H. Nájera, R. Gutiérrez, R. González, C. García, R. Méndez, M. Rojas. (2016). Degradation of gestodene (GES)–17α-ethinylestradiol (EE2) mixture by electrochemical oxidation. Journal of Water and Health. (2016), 14 (6), 980–988.
- K. Rajeshwar, J. Ibanez. Environmental electrochemistry: Fundamentals and applications in pollution sensors and abatement. Academic Press. (1997) Chapter 7.
- B. Boye, E. Brillas, B. Marselli, P.A. Michaud, C. Comninellis, G. Farnia, G. Sandonà. Electrochemical incineration of chloromethylphenoxy herbicides in acid medium by anodic oxidation with boron-doped diamond electrode. Electrochimica Acta. (2006), 51 (14), pp. 2872–2880.


